Aniracetam
A fat-soluble nootropic compound of the racetam class, studied for its potential effects on memory, cognition, and anxiety through AMPA receptor modulation.
Aniracetam is a fat-soluble racetam nootropic known for its anxiolytic and cognitive-enhancing properties. It modulates AMPA receptors and cholinergic transmission, potentially improving memory, learning, and reducing anxiety.
Overview
Aniracetam (1-p-anisoyl-2-pyrrolidinone) is a fat-soluble ampakine nootropic in the racetam class of compounds, first synthesized in the 1970s by Hoffmann-La Roche. Unlike its hydrophilic predecessor piracetam, aniracetam's lipophilicity allows it to cross the blood-brain barrier rapidly, reaching peak brain concentrations within 20–30 minutes of oral administration. It is marketed as a prescription drug in parts of Europe and Japan under trade names including Ampamet and Draganon, while in the United States it is sold as an unregulated dietary supplement.
Aniracetam's primary mechanism involves positive allosteric modulation of AMPA receptors, a subtype of ionotropic glutamate receptor critical for synaptic plasticity, learning, and memory. By slowing the desensitization of these receptors, it enhances excitatory neurotransmission. Additionally, aniracetam modulates the cholinergic system by promoting acetylcholine release, and influences dopaminergic and serotonergic pathways, which may account for its reported anxiolytic effects. Its major metabolite, N-anisoyl-GABA (70–80% of metabolism), also contributes to its pharmacological profile.
Preclinical studies in cognitively impaired animal models have demonstrated improvements in memory acquisition, spatial learning, and anxiety-related behavior. However, evidence in healthy subjects is limited; controlled studies in normal mice have failed to detect significant cognitive enhancement. Aniracetam may also support neuroprotection through increased BDNF expression and antioxidant activity. While it remains popular in the nootropic community, rigorous large-scale human clinical trials are sparse, and its full mechanism of action is not completely understood.
Mechanism of Action
AMPA Receptor Positive Allosteric Modulation
Aniracetam (1-p-anisoyl-2-pyrrolidinone) is a lipophilic pyrrolidinone nootropic that primarily acts as a positive allosteric modulator (PAM) of AMPA-type glutamate receptors. It binds at the dimer interface of AMPA receptor subunits (GluA1-4), stabilizing the receptor in its active conformation and slowing both desensitization and deactivation kinetics. This prolongs excitatory postsynaptic currents (EPSCs) and enhances glutamatergic neurotransmission without direct agonist activity (PMID: 8395949).
Metabotropic Glutamate Receptor Effects
Aniracetam also acts on group I metabotropic glutamate receptors (mGluR1/5), potentiating their Gq-coupled signaling. This enhances phospholipase C (PLC) activation, IP3-mediated intracellular calcium release, and downstream protein kinase C (PKC) activity, further supporting synaptic plasticity and long-term potentiation (LTP) (PMID: 11597608).
Cholinergic & Dopaminergic Modulation
Through its AMPA-mediated effects on cortical circuits, aniracetam secondarily increases acetylcholine release in the prefrontal cortex and hippocampus via glutamate-driven activation of cholinergic projection neurons. It also modulates dopamine and serotonin release in the prefrontal cortex, amygdala, and striatum, contributing to its anxiolytic and mood-enhancing properties (PMID: 11412837).
BDNF & Synaptic Plasticity
Aniracetam-enhanced AMPA receptor signaling drives calcium influx that activates CaMKII and CREB phosphorylation, upregulating brain-derived neurotrophic factor (BDNF) expression. BDNF in turn promotes dendritic spine growth, synaptogenesis, and the consolidation of LTP — the cellular correlate of memory formation (PMID: 10996082).
Metabolic Profile
Aniracetam is rapidly metabolized (t½ ~35 min) to active metabolites including N-anisoyl-GABA and p-anisic acid, which contribute to anxiolytic effects via GABAergic and dopaminergic modulation respectively.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
Research
Reported Effects
Quick Onset:: Effects typically noticed within 30-60 minutes.. Short Half-Life:: Requires multiple daily doses due to 1-2 hour half-life.. Requires Choline:: Best effects when stacked with a choline source.
- Effects typically noticed within 30-60 minutes.
- Requires multiple daily doses due to 1-2 hour half-life.
- Best effects when stacked with a choline source.
Safety Profile
Common Side Effects
- Headache, dizziness, and insomnia
- Gastrointestinal disturbances (nausea, diarrhea, stomach pain)
- Anxiety, irritability, and restlessness at higher doses
- Mild skin rash in sensitive individuals
Serious Concerns
- Aniracetam is not FDA-approved in the United States and is sold as an unregulated supplement. It is a prescription drug in some European countries (e.g., under the brand name Draganon in Japan, though discontinued).
- Long-term safety data in humans is limited. Most studies are short-term (weeks to months).
- Rare reports of vertigo and psychomotor agitation.
Contraindications
- Known hypersensitivity to aniracetam or other racetams
- Severe renal impairment (primary renal excretion of metabolites)
- Severe hepatic impairment (hepatic metabolism via CYP enzymes)
- Pregnancy and breastfeeding (no safety data)
- Huntington disease (theoretical concern with glutamatergic modulation)
Drug Interactions
- Anticoagulants (warfarin): Racetams may potentiate anticoagulant effects; monitor INR
- CNS depressants (benzodiazepines, alcohol): Potential for altered sedation profiles
- Cholinergic drugs: Additive cholinergic activity when stacked with choline sources or acetylcholinesterase inhibitors
- Other racetams/nootropics: Unpredictable synergistic or antagonistic interactions
Special Populations
- Pediatric: No established safety or efficacy; not recommended
- Geriatric: Some clinical research in elderly dementia patients; use with caution and medical supervision
- Hepatic/renal impairment: Dose adjustment likely needed; no formal guidelines exist
Monitoring
- Cognitive and mood assessment during use
- Renal and hepatic function tests periodically
- Monitor for headache, which may indicate choline depletion (often managed by co-supplementing with a choline source)
Pharmacokinetic Profile
Aniracetam — Pharmacokinetic Curve
SubcutaneousQuick Start
- Typical Dose
- 750-1500mg daily, divided into 2-3 doses.
Molecular Structure
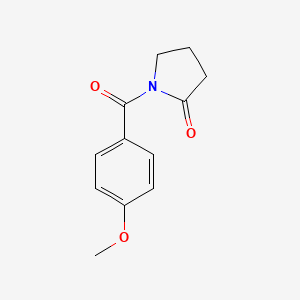
- Formula
- C12H13NO3
- Weight
- 219.24 Da
- PubChem CID
- 2196
- Exact Mass
- 219.0895 Da
- LogP
- 1.6
- TPSA
- 46.6 Ų
- H-Bond Donors
- 0
- H-Bond Acceptors
- 3
- Rotatable Bonds
- 2
- Complexity
- 282
Identifiers (SMILES, InChI)
InChI=1S/C12H13NO3/c1-16-10-6-4-9(5-7-10)12(15)13-8-2-3-11(13)14/h4-7H,2-3,8H2,1H3
ZXNRTKGTQJPIJK-UHFFFAOYSA-NSafety Profile
Common Side Effects
- Headaches:: Common if not taken with adequate choline.
- Digestive Issues:: Some users report mild GI discomfort.
- Generally Mild:: Side effects are typically minimal at normal doses.
References (10)
- [4]Comparative kinetic studies and pH-rate profiling of aniracetam degradation
→ Develops HPLC methodology for measuring aniracetam stability across varying pH conditions.
- [10]Determination of aniracetam's main metabolite, N-anisoyl-GABA, in human plasma by LC–MS/MS
→ Develops efficient method for measuring aniracetam's primary metabolite in pharmacokinetic studies.
- [2]Aniracetam: An overview of its pharmacodynamic and pharmacokinetic properties
→ Reviews aniracetam's cognitive enhancement effects in elderly patients with Alzheimer's-related impairment.
- [3]Treatment of insomnia by concomitant therapy with Zopiclone and Aniracetam
→ Combined Zopiclone and Aniracetam achieved 50%+ sleep duration increases in elderly patients.
- [5]Aniracetam and DNQX affect the acquisition of rapid tolerance to ethanol in mice
→ Investigates AMPA receptor involvement in ethanol tolerance development.
- [6]Sensitive and selective LC–MS/MS method for the quantification of aniracetam in human plasma
→ Develops method for measuring aniracetam plasma levels with 0.2 ng/mL detection limit.
- [7]Postnatal aniracetam treatment improves prenatal ethanol induced attenuation of AMPA receptor-mediated synaptic transmission
→ Demonstrates postnatal aniracetam reverses prenatal alcohol-induced cognitive deficits.
- [8]Development and validation of a liquid chromatographic method for the simultaneous determination of aniracetam
→ Creates HPLC method with UV detection for identifying aniracetam and impurities.
- [9]Ameliorating effects of preadolescent aniracetam treatment on prenatal ethanol-induced impairment
→ Shows aniracetam improves synaptic transmission and cognitive potential after prenatal ethanol exposure.
- [1]Aniracetam: its novel therapeutic potential in cerebral dysfunctional disorders
→ Examines aniracetam's potential for treating CNS disorders using animal models.