Magnesium Citrate
A highly bioavailable magnesium salt bound to citric acid, widely used for general magnesium repletion, bowel regularity, and as a gentle osmotic laxative at higher doses.
Magnesium citrate is an organic magnesium salt that combines magnesium with citric acid, commonly used as a dietary supplement and occasional laxative. It works by increasing magnesium bioavailability in the body, supporting over 300 enzymatic reactions including energy production, nerve function, muscle contraction, and blood pressure regulation. Due to its osmotic laxative properties, it also draws water into the intestines to promote bowel movements.
Overview
Magnesium citrate is a magnesium salt formed by combining elemental magnesium with citric acid, providing approximately 16% elemental magnesium by weight. It is one of the most widely used and well-studied magnesium supplementation forms, valued for its superior bioavailability compared to magnesium oxide (the most common but least bioavailable form) — randomized crossover studies demonstrate 25–30% greater absorption from citrate versus oxide. The citrate anion itself contributes to the compound's clinical utility: citrate is readily metabolized in the Krebs cycle and alkalinizes urine, making magnesium citrate particularly useful for kidney stone prevention (it increases urinary citrate excretion, which inhibits calcium oxalate and calcium phosphate crystal formation). At standard supplemental doses (200–400 mg elemental magnesium), magnesium citrate corrects deficiency and supports the enzyme's 300+ enzymatic functions; at higher doses (1–3 g of the salt), it acts as an osmotic laxative.
Magnesium deficiency affects an estimated 50–80% of the Western population, driven by soil mineral depletion, processed food consumption, chronic stress (which increases urinary magnesium excretion), and medications (proton pump inhibitors, diuretics, certain antibiotics). Subclinical deficiency manifests as muscle cramps, insomnia, anxiety, cardiac arrhythmias, migraines, and insulin resistance — symptoms often attributed to other causes. Magnesium citrate effectively repletes intracellular magnesium stores, supporting ATP production (magnesium is required for all ATP-dependent reactions), neuromuscular function, blood pressure regulation (through endothelial-dependent vasodilation and calcium channel antagonism), and glucose metabolism. Clinical trials demonstrate that magnesium supplementation (300–600 mg/day elemental) reduces blood pressure by 3–4 mmHg systolic, decreases migraine frequency by 40–50%, and improves insulin sensitivity in metabolic syndrome.
The choice among magnesium forms should be guided by the primary clinical goal. While magnesium citrate is an excellent general-purpose form, magnesium-glycinate (bisglycinate) may be preferred for sleep and anxiety due to glycine's co-agonist activity at NMDA receptors and inhibitory neurotransmitter role. Magnesium-l-threonate specifically elevates brain magnesium levels and is targeted at cognitive function and neuroplasticity. Magnesium-taurate combines magnesium with the cardioprotective amino acid taurine for cardiovascular applications. Magnesium citrate pairs well with vitamin-d3 (magnesium is required for vitamin D activation and metabolism), vitamin-b6 (which enhances intracellular magnesium accumulation), potassium for comprehensive electrolyte support, and l-citrulline for combined vascular relaxation. Typical supplemental doses of 200–400 mg elemental magnesium daily are well-tolerated, with loose stools being the primary dose-limiting side effect — a property that makes magnesium citrate the preferred form for individuals with concurrent constipation.
Mechanism of Action
Magnesium citrate is a salt of elemental magnesium bound to citric acid, providing approximately 16% elemental magnesium by weight. The citrate anion significantly enhances magnesium bioavailability compared to inorganic forms (oxide, sulfate) through two mechanisms: first, citrate maintains magnesium solubility in the alkaline environment of the small intestine by chelation, preventing precipitation as insoluble magnesium hydroxide; second, citrate enhances paracellular absorption by transiently opening tight junctions through calcium chelation at the claudin level, allowing magnesium to pass between enterocytes. Citrate itself is absorbed and enters the tricarboxylic acid (TCA) cycle as a metabolic intermediate, providing additional metabolic substrate.
Once absorbed, magnesium serves as an essential cofactor for over 300 enzymatic reactions, most critically those involving ATP. Magnesium forms a complex with ATP (Mg-ATP) that is the actual substrate recognized by kinases, ATPases, and other phosphotransferases. Without adequate magnesium, ATP cannot be properly utilized for cellular energy transfer. In the nervous system, magnesium occupies the voltage-dependent binding site within the NMDA receptor channel pore, acting as a physiological channel blocker at resting membrane potential. This voltage-dependent block must be relieved by depolarization before calcium can flow through the channel, making magnesium a critical regulator of excitatory neurotransmission, synaptic plasticity, and excitotoxic injury prevention.
Magnesium also regulates calcium homeostasis by competing with calcium for binding sites on proteins and channels, including L-type voltage-gated calcium channels and ryanodine receptors in muscle tissue. Adequate magnesium maintains normal cardiac rhythm by stabilizing the resting membrane potential and modulating repolarization currents. In bone metabolism, magnesium is essential for parathyroid hormone (PTH) secretion and for converting vitamin D to its active 1,25-dihydroxyvitamin D3 form via renal 1-alpha-hydroxylase. The osmotic properties of unabsorbed citrate in the intestinal lumen draw water into the bowel via osmosis, providing the mild laxative effect for which magnesium citrate is commonly used, making it particularly suitable for individuals with constipation.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
Research
Reported Effects
Bioavailability:: Consistently praised as one of the most absorbable forms of magnesium, superior to oxide and comparable to glycinate. Dose-Dependent Response:: Effectiveness varies by individual needs; deficient users report more dramatic improvements than those with adequate baseline levels. Timing Matters:: Most effective when taken in evening for sleep benefits, though some prefer splitting doses throughout the day. Individual Variation:: Response varies significantly based on baseline magnesium status, diet, and specific health conditions being addressed
- Consistently praised as one of the most absorbable forms of magnesium, superior to oxide and comparable to glycinate
- Effectiveness varies by individual needs; deficient users report more dramatic improvements than those with adequate baseline levels
- Most effective when taken in evening for sleep benefits, though some prefer splitting doses throughout the day
- Response varies significantly based on baseline magnesium status, diet, and specific health conditions being addressed
Safety Profile
Magnesium citrate can cause gastrointestinal side effects like diarrhea, cramping, and nausea, especially at higher doses. People with kidney problems, bowel obstructions, or certain heart conditions should avoid it, as impaired kidney function can lead to dangerous hypermagnesemia. It may interact with antibiotics, heart medicines, and other medications.
Pharmacokinetic Profile
Quick Start
- Typical Dose
- Most users take 200-400mg of elemental magnesium daily, with 300mg being the most commonly reported effective dose
Molecular Structure
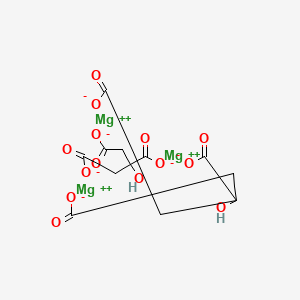
- Formula
- C12H10Mg3O14
- Weight
- 451.12 Da
- PubChem CID
- 6099959
- Exact Mass
- 449.9622 Da
- TPSA
- 281 Ų
- H-Bond Donors
- 2
- H-Bond Acceptors
- 14
- Rotatable Bonds
- 4
- Complexity
- 211
Identifiers (SMILES, InChI)
InChI=1S/2C6H8O7.3Mg/c2*7-3(8)1-6(13,5(11)12)2-4(9)10;;;/h2*13H,1-2H2,(H,7,8)(H,9,10)(H,11,12);;;/q;;3*+2/p-6
PLSARIKBYIPYPF-UHFFFAOYSA-HSafety Profile
Common Side Effects
- Digestive Issues:: Most common side effect is loose stools or diarrhea, particularly at doses above 400mg or when taken on empty stomach
- Initial Adjustment:: Some users experience temporary stomach discomfort, bloating, or cramping during first few days of supplementation
- Individual Tolerance:: Laxative effect threshold varies significantly between individuals; some tolerate 600mg while others react to 200mg
- Timing-Related:: Taking with food typically reduces digestive side effects, though may slightly decrease absorption rate
References (2)
- [2]Effects of Magnesium Citrate, Magnesium Oxide, and Magnesium Sulfate Supplementation on Arterial Stiffness: A Randomized, Double-Blind, Placebo-Controlled Intervention Trial
→ 24-week supplementation with 450mg/day magnesium citrate did not significantly affect arterial stiffness or blood pressure in overweight subjects, though it effectively increased plasma and urinary magnesium levels.
- [1]Higher bioavailability of magnesium citrate as compared to magnesium oxide shown by evaluation of urinary excretion and serum levels after single-dose administration in a randomized cross-over study
→ Magnesium citrate demonstrated significantly higher bioavailability compared to magnesium oxide, with greater urinary excretion and serum levels following single-dose administration in healthy subjects.