L-DOPA
The direct biosynthetic precursor to dopamine and the gold-standard treatment for Parkinson's disease, also found naturally in Mucuna pruriens (velvet bean).
L-DOPA (levodopa) is a naturally occurring amino acid that serves as the direct precursor to dopamine, a crucial neurotransmitter involved in movement, motivation, and mood regulation. It is the gold standard treatment for Parkinson's disease, as it can cross the blood-brain barrier and be converted into dopamine to replace depleted levels. L-DOPA is also found naturally in plants like Mucuna pruriens (velvet bean) and is sometimes used as a supplement, though this practice carries significant risks.
Overview
L-DOPA (levodopa, L-3,4-dihydroxyphenylalanine) is an amino acid intermediate in the catecholamine biosynthetic pathway, serving as the immediate precursor to dopamine through decarboxylation by aromatic L-amino acid decarboxylase (AADC). As the most effective treatment for Parkinson's disease since its introduction in the 1960s, L-DOPA crosses the blood-brain barrier (unlike dopamine itself) and is converted to dopamine in surviving nigrostriatal neurons, directly replenishing the neurotransmitter deficit underlying parkinsonian motor symptoms. Pharmaceutical formulations combine L-DOPA with peripheral decarboxylase inhibitors (carbidopa or benserazide) to prevent premature conversion to dopamine outside the CNS, improving bioavailability and reducing peripheral side effects such as nausea and hypotension.
Beyond Parkinson's disease, L-DOPA is naturally present in significant quantities in mucuna-pruriens (velvet bean), which contains approximately 3.6–4.2% L-DOPA by dry weight in its seeds. Mucuna-based supplementation has gained attention in both traditional Ayurvedic medicine and modern biohacking communities for mood enhancement, motivation, libido support, and growth hormone secretion. L-DOPA stimulates pituitary growth hormone release — a property that has made it a component of some growth hormone stimulation test protocols. It also serves as a precursor to norepinephrine and epinephrine through sequential enzymatic steps, giving it broad influence over catecholaminergic function, reward processing, and stress response.
However, L-DOPA supplementation requires careful consideration. Long-term use in Parkinson's patients eventually leads to motor complications including dyskinesias and "wearing-off" phenomena, reflecting dopaminergic neuronal degeneration and altered receptor sensitivity. Even in supplement form via Mucuna pruriens, chronic high-dose use can suppress endogenous dopamine synthesis through negative feedback and deplete serotonin (since AADC is shared between dopamine and serotonin pathways). For dopaminergic support without direct precursor loading, alternatives include l-tyrosine (which provides upstream substrate), bromantane (which upregulates tyrosine hydroxylase), and phenylpiracetam for dopaminergic modulation. Users considering L-DOPA supplementation should do so under clinical supervision, particularly at doses exceeding those in typical Mucuna extracts.
Mechanism of Action
L-DOPA (levodopa, L-3,4-dihydroxyphenylalanine) is the immediate biosynthetic precursor to dopamine and crosses the blood-brain barrier via the large neutral amino acid transporter (LAT1/SLC7A5), unlike dopamine itself which cannot penetrate the BBB. Once in the central nervous system, L-DOPA is decarboxylated by aromatic L-amino acid decarboxylase (AADC, also known as DOPA decarboxylase) to produce dopamine. This conversion occurs in both surviving dopaminergic neurons in the substantia nigra pars compacta and in serotonergic neurons, glial cells, and other AADC-expressing cells throughout the brain. The newly synthesized dopamine is packaged into synaptic vesicles by vesicular monoamine transporter 2 (VMAT2) and released into the synaptic cleft, where it activates dopamine receptors (D1-D5) on postsynaptic neurons in the striatum and other target regions.
In Parkinson's disease, the progressive degeneration of nigrostriatal dopaminergic neurons results in severe striatal dopamine depletion, leading to the cardinal motor symptoms of bradykinesia, rigidity, tremor, and postural instability. The striatal dopaminergic system modulates the balance between the direct pathway (D1 receptor-mediated, facilitating movement) and the indirect pathway (D2 receptor-mediated, inhibiting movement) of the basal ganglia motor circuit. Dopamine depletion shifts this balance toward excessive indirect pathway activity, resulting in increased inhibitory output from the globus pallidus internus (GPi) and substantia nigra pars reticulata (SNr) to the thalamus, suppressing thalamocortical motor drive. L-DOPA supplementation restores striatal dopamine levels, rebalancing direct and indirect pathway activity and alleviating motor symptoms.
Peripherally, L-DOPA is rapidly decarboxylated to dopamine by AADC in the gut and liver, and O-methylated by catechol-O-methyltransferase (COMT) to 3-O-methyldopa. These peripheral conversions reduce CNS bioavailability to approximately 1-5% of an oral dose. For this reason, L-DOPA is almost always co-administered with a peripheral AADC inhibitor (carbidopa or benserazide) that does not cross the BBB, blocking peripheral dopamine formation and dramatically increasing the fraction reaching the brain. COMT inhibitors (entacapone, tolcapone) may be added to further reduce peripheral L-DOPA metabolism. The pulsatile nature of oral L-DOPA delivery, with fluctuating plasma and brain levels, eventually contributes to motor complications including wearing-off phenomena and dyskinesias, driven by non-physiological intermittent stimulation of dopamine receptors and downstream plasticity changes in striatal medium spiny neurons involving alterations in DARPP-32 signaling, glutamatergic NMDA receptor sensitization, and aberrant FosB/DeltaFosB transcription factor accumulation.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
Research
Reported Effects
Medical Use vs Supplementation:: L-DOPA is highly effective for Parkinson's disease when properly prescribed with carbidopa, but over-the-counter supplements lack this crucial component and are largely ineffective or dangerous. Downregulation Risk:: Users and experts consistently warn that L-DOPA supplementation can suppress natural tyrosine hydroxylase expression and dopamine synthesis, making recovery difficult and symptoms worse long-term. Better Alternatives Exist:: Community consensus strongly favors L-tyrosine or N-acetyl-L-tyrosine as safer precursors that allow the body to regulate dopamine production naturally. Short-Term Only:: Those who found any benefit emphasized cycling and very short-term use (days to weeks maximum), never continuous supplementation
- L-DOPA is highly effective for Parkinson's disease when properly prescribed with carbidopa, but over-the-counter supplements lack this crucial component and are largely ineffective or dangerous
- Users and experts consistently warn that L-DOPA supplementation can suppress natural tyrosine hydroxylase expression and dopamine synthesis, making recovery difficult and symptoms worse long-term
- Community consensus strongly favors L-tyrosine or N-acetyl-L-tyrosine as safer precursors that allow the body to regulate dopamine production naturally
- Those who found any benefit emphasized cycling and very short-term use (days to weeks maximum), never continuous supplementation
Safety Profile
Safety Profile: L-DOPA (Levodopa)
Common Side Effects
- Nausea and vomiting (very common, especially early in treatment; mitigated by co-administration with carbidopa or benserazide)
- Orthostatic hypotension: dizziness, lightheadedness, and fainting upon standing
- Drowsiness and somnolence (including sudden-onset sleep episodes)
- Dyskinesias: involuntary movements (choreoathetoid) that develop with chronic use, affecting up to 40–80% of patients after 5–10 years
- Dry mouth, anorexia, and weight changes
- Darkening of urine, sweat, and saliva (harmless but alarming to patients)
- Vivid dreams and insomnia
Serious Adverse Effects
- Motor fluctuations ("on-off" phenomenon): unpredictable switching between mobility and immobility with chronic use
- Psychiatric effects: visual hallucinations, paranoia, psychosis, confusion, and agitation (more common in elderly and with advancing disease)
- Impulse control disorders: pathological gambling, hypersexuality, compulsive shopping, and binge eating (associated with dopaminergic stimulation)
- Neuroleptic malignant syndrome (NMS): rare, life-threatening reaction upon abrupt withdrawal — hyperthermia, rigidity, altered consciousness, autonomic instability
- Melanoma risk: epidemiological association with increased melanoma incidence in Parkinson's patients on levodopa (causality debated)
- Cardiac arrhythmias (rare, particularly with non-selective DOPA decarboxylase inhibitor use)
- Severe depression and suicidal ideation
Contraindications
- Concurrent non-selective MAO inhibitors (phenelzine, tranylcypromine): risk of hypertensive crisis — must be discontinued 2 weeks prior
- Narrow-angle glaucoma (may increase intraocular pressure)
- Known hypersensitivity to levodopa or formulation components
- Pheochromocytoma (catecholamine surge risk)
- History of melanoma or suspicious undiagnosed skin lesions
- Severe psychotic disorders not controlled by medication
Drug Interactions
- Non-selective MAO inhibitors: CONTRAINDICATED — hypertensive crisis risk
- Antipsychotics (haloperidol, risperidone, olanzapine): antagonize dopaminergic effects; worsen parkinsonism
- Metoclopramide and prochlorperazine: dopamine antagonists that reduce L-DOPA efficacy
- Pyridoxine (vitamin B6): enhances peripheral DOPA decarboxylation; reduces efficacy unless carbidopa is co-administered
- Iron supplements: chelate levodopa in the GI tract, reducing absorption by 30–50%; separate by 2+ hours
- High-protein meals: compete with levodopa for intestinal and blood-brain barrier amino acid transporters; may reduce efficacy
- Selegiline/rasagiline (MAO-B inhibitors): generally safe but may potentiate dopaminergic side effects
- Antihypertensives: additive hypotension
Population-Specific Considerations
- Pregnancy: Category C; teratogenic in animal studies; use only when benefit clearly outweighs risk
- Lactation: levodopa inhibits prolactin secretion and may suppress lactation; excreted in breast milk
- Children: rarely used in pediatric populations; approved for specific dystonia types under specialist supervision
- Elderly: increased susceptibility to psychiatric effects, orthostatic hypotension, and falls; start at lowest dose with slow titration
- Renal/Hepatic impairment: dose adjustment may be necessary; monitor closely
Pharmacokinetic Profile
Molecular Structure
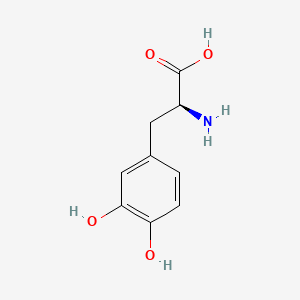
- Formula
- C9H11NO4
- Weight
- 197.19 Da
- PubChem CID
- 6047
- Exact Mass
- 197.0688 Da
- LogP
- -2.7
- TPSA
- 104 Ų
- H-Bond Donors
- 4
- H-Bond Acceptors
- 5
- Rotatable Bonds
- 3
- Complexity
- 209
Identifiers (SMILES, InChI)
InChI=1S/C9H11NO4/c10-6(9(13)14)3-5-1-2-7(11)8(12)4-5/h1-2,4,6,11-12H,3,10H2,(H,13,14)/t6-/m0/s1
WTDRDQBEARUVNC-LURJTMIESA-NSafety Profile
Common Side Effects
- Dyskinesia:: Multiple users reported involuntary movements, head jerking, tremors, and coordination problems, sometimes appearing rapidly after starting supplementation
- Nausea and Gastrointestinal Issues:: Common reports of nausea, vomiting, and digestive discomfort, particularly when taken without food or carbidopa
- Withdrawal Effects:: Users describe severe crashes, worsened depression, anhedonia, and anxiety when discontinuing L-DOPA supplements after regular use
- Dangerous Drug Interactions:: Serious interactions reported with antidepressants (especially Wellbutrin, SSRIs), causing serotonin syndrome symptoms and amplified side effects
References (6)
- [1]Parkinson's disease: A review
→ Levodopa remains the mainstay treatment for Parkinson's disease motor symptoms, though long-term use commonly leads to levodopa-induced dyskinesia (involuntary movements), which can occur after extended treatment or sometimes even within days to months.
- [2]L-DOPA ameliorates hippocampus-based mitochondria respiratory dysfunction caused by GCI/R injury
→ L-DOPA showed neuroprotective effects by improving mitochondrial respiratory function in the hippocampus following global cerebral ischemia/reperfusion injury in animal models.
- [3]Dietary Approaches to Improve Efficacy and Control Side Effects of Levodopa Therapy in Parkinson's Disease: A Systematic Review
→ Diet plays a crucial role in levodopa therapy effectiveness, as protein intake can interfere with absorption, and nutritional approaches may help manage motor fluctuations and side effects in Parkinson's patients.
- [4]How to Optimize the Effectiveness and Safety of Parkinson's Disease Therapy? - A Systematic Review of Drugs Interactions with Food and Dietary Supplements
→ Evidence suggests positive interactions between levodopa and coffee, fiber, and vitamin C, while proper timing of medication intake relative to food can significantly optimize Parkinson's disease treatment outcomes.
- [5]Neuroprotective effect of Bacillus subtilis in haloperidol induced rat model, targeting the microbiota-gut-brain axis
→ Study compared probiotics with L-DOPA as standard treatment in a Parkinson's model, demonstrating the importance of the gut-brain axis in dopaminergic function and potential therapeutic approaches.
- [6]Overview of Parkinson's disease
→ Parkinson's disease involves loss of dopaminergic neurons in the substantia nigra, and while levodopa effectively manages motor symptoms and forms the basis for clinical diagnosis, there is no cure and long-term management presents significant challenges.