Phenylalanine
An essential aromatic amino acid that serves as a precursor to tyrosine, dopamine, norepinephrine, and epinephrine, playing a critical role in neurotransmitter synthesis, mood regulation, and pain modulation.
Phenylalanine is an essential amino acid that serves as a precursor to tyrosine and subsequently dopamine, norepinephrine, and epinephrine. It exists in three forms: L-phenylalanine (natural form found in proteins), D-phenylalanine (synthetic form that may inhibit endorphin breakdown), and DL-phenylalanine (DLPA, a combination of both). It is primarily used as a dietary supplement for mood support, ADHD symptoms, focus enhancement, and pain management through its role in neurotransmitter synthesis.
Overview
Phenylalanine is one of the nine essential amino acids that must be obtained through diet, as humans lack the enzymatic machinery for its de novo synthesis. It exists in two enantiomeric forms: L-phenylalanine, the naturally occurring form found in dietary protein, and D-phenylalanine, a synthetic mirror image with distinct pharmacological properties. A racemic mixture (DL-phenylalanine or DLPA) is also used therapeutically. L-phenylalanine is hydroxylated by phenylalanine hydroxylase (PAH) to produce L-tyrosine, which then feeds into the catecholamine biosynthesis pathway generating L-DOPA, dopamine, norepinephrine, and epinephrine. This cascade makes phenylalanine a foundational upstream precursor for neurotransmitters that regulate mood, motivation, attention, and the stress response.
D-phenylalanine operates through a different mechanism, primarily inhibiting enkephalinase — the enzyme responsible for degrading endogenous opioid peptides (enkephalins). By prolonging the activity of met-enkephalin and leu-enkephalin, D-phenylalanine may enhance the body's intrinsic pain-modulating systems, which has generated interest in its use for chronic pain management and as an adjunct in pain therapy protocols. DLPA supplements are often marketed for combined mood and analgesic support, leveraging both enantiomers' distinct actions.
Clinically, phenylalanine is most notable in the context of phenylketonuria (PKU), an inborn error of metabolism caused by PAH deficiency that leads to toxic phenylalanine accumulation. Outside of PKU, supplemental phenylalanine has been investigated for depression — particularly in cases involving catecholamine depletion — and for vitiligo, where it may support melanin production in conjunction with UVA phototherapy. Phenylalanine is abundant in high-protein foods including eggs, dairy, meat, soy, and is also a component of the artificial sweetener aspartame. It works synergistically with vitamin B6, folate, and iron as cofactors required for its enzymatic conversion to downstream neurotransmitters.
Mechanism of Action
Mechanism of Action
Phenylalanine is one of the nine essential amino acids that must be obtained from diet. It exists as L-phenylalanine (natural dietary form) and D-phenylalanine (synthetic enantiomer), with DL-phenylalanine supplements containing both forms for complementary effects.
Catecholamine Precursor Pathway
The primary biochemical role of L-phenylalanine is as the upstream precursor for catecholamine neurotransmitter synthesis. Phenylalanine hydroxylase (PAH), a hepatic enzyme requiring tetrahydrobiopterin (BH4) and molecular oxygen, converts phenylalanine to tyrosine. Tyrosine then enters the catecholamine pathway: tyrosine hydroxylase (TH) produces L-DOPA, aromatic amino acid decarboxylase (AADC) produces dopamine, dopamine β-hydroxylase (DBH) produces norepinephrine, and phenylethanolamine N-methyltransferase (PNMT) produces epinephrine. Supplemental phenylalanine may increase catecholamine synthesis when precursor availability is rate-limiting.
D-Phenylalanine and Endogenous Opioids
D-phenylalanine has a distinct mechanism: it inhibits carboxypeptidase A and enkephalinase, the enzymes responsible for degrading endogenous opioid peptides (met-enkephalin, leu-enkephalin). By slowing enkephalin catabolism, D-phenylalanine extends the duration and intensity of endogenous analgesic signaling. This has been investigated for chronic pain conditions where endogenous opioid tone is depleted.
Trace Amine Signaling
Phenylalanine decarboxylation produces β-phenylethylamine (PEA), a trace amine neurotransmitter that activates trace amine-associated receptor 1 (TAAR1). TAAR1 activation modulates dopaminergic and serotonergic neurotransmission, and PEA itself triggers rapid dopamine and norepinephrine release from presynaptic terminals, producing acute mood-elevating and stimulant effects.
Clinical Relevance
Phenylalanine deficiency impairs catecholamine synthesis, contributing to depression, fatigue, and cognitive impairment. Conversely, PAH deficiency causes phenylketonuria (PKU), where toxic phenylalanine accumulation damages the developing brain. The therapeutic window for phenylalanine supplementation exists between deficiency states and the toxic accumulation seen in PKU.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
Research
Reported Effects
Highly Individual Response:: Effectiveness appears strongly dependent on individual biochemistry, with some users experiencing dramatic benefits while others notice nothing at all, possibly related to COMT gene variants affecting phenylalanine breakdown. Form Matters:: DL-phenylalanine (DLPA) reported more effective than L-phenylalanine alone, with the D-form contributing endorphin-related benefits; typical reported effective dose is 500-1500mg. Requires Cofactors:: Users emphasize importance of taking with B-vitamins (especially B6) and vitamin C for proper conversion to neurotransmitters; without cofactors, effectiveness diminishes. Tolerance and Cycling:: Some users report diminishing effects with continuous use, recommending cycling or occasional use rather than daily supplementation to maintain effectiveness
- Effectiveness appears strongly dependent on individual biochemistry, with some users experiencing dramatic benefits while others notice nothing at all, possibly related to COMT gene variants affecting phenylalanine breakdown
- DL-phenylalanine (DLPA) reported more effective than L-phenylalanine alone, with the D-form contributing endorphin-related benefits; typical reported effective dose is 500-1500mg
- Users emphasize importance of taking with B-vitamins (especially B6) and vitamin C for proper conversion to neurotransmitters; without cofactors, effectiveness diminishes
- Some users report diminishing effects with continuous use, recommending cycling or occasional use rather than daily supplementation to maintain effectiveness
Safety Profile
Safety Profile: Phenylalanine
Common Side Effects
- Headache and migraine exacerbation in susceptible individuals
- Nausea, heartburn, and gastrointestinal discomfort
- Anxiety, jitteriness, and nervousness (particularly D- and DL-phenylalanine)
- Insomnia when taken late in the day
- Mild hypertension at high doses
- Worsening of tardive dyskinesia symptoms
Serious Adverse Effects
- Phenylketonuria (PKU) crisis: Severe neurotoxicity in individuals with PKU who cannot metabolize phenylalanine; intellectual disability, seizures, and brain damage with chronic excess intake
- Exacerbation of schizophrenia symptoms (increases dopamine and norepinephrine)
- Hypertensive crisis when combined with MAO inhibitors
- Severe allergic reactions (rare)
- Potential interference with levodopa absorption and efficacy
Contraindications
- Phenylketonuria (PKU): Absolute contraindication; phenylalanine supplementation is strictly prohibited
- Carriers of PKU (heterozygotes) should use with caution
- Concurrent use of MAO inhibitors (risk of hypertensive crisis)
- Schizophrenia or psychotic disorders (may worsen symptoms)
- Pregnancy (excess phenylalanine is teratogenic; particularly critical for maternal PKU)
- Melanoma or history of melanoma (phenylalanine is a precursor to melanin)
- Tardive dyskinesia
Drug Interactions
- MAO inhibitors (phenelzine, tranylcypromine): Dangerous hypertensive crisis; phenylalanine increases catecholamine synthesis
- Levodopa/carbidopa: Competition for amino acid transport; may reduce levodopa efficacy; separate dosing by 2+ hours
- Antipsychotics: May counteract therapeutic effects by increasing dopamine precursors
- Antihypertensives: High-dose phenylalanine may elevate blood pressure, reducing drug efficacy
- Baclofen: May reduce phenylalanine absorption
- Other amino acid supplements: Competition for transport across blood-brain barrier
Population-Specific Considerations
- Depression (DL-phenylalanine): Used as adjunctive therapy; typical doses 500-1,500 mg/day; may take 2-4 weeks for effect
- Chronic pain (DL-phenylalanine): D-form inhibits enkephalinase, prolonging endorphin activity; doses up to 2,000 mg/day studied
- PKU screening: All individuals should confirm they do not have PKU before supplementing
- Pregnant women with PKU: Maternal phenylalanine levels must be strictly controlled to prevent fetal damage
- Children: Not recommended without medical supervision; especially important to rule out PKU
Pharmacokinetic Profile
Quick Start
- Typical Dose
- Most users report taking 500-1500mg, with 500mg being common starting point and 1500mg on higher end; typically taken on empty stomach in morning
Molecular Structure
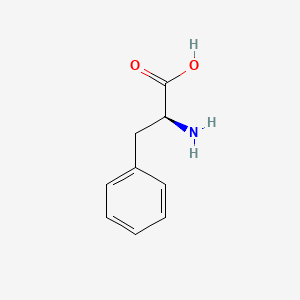
- Formula
- C9H11NO2
- Weight
- 165.19 Da
- PubChem CID
- 6140
- Exact Mass
- 165.0790 Da
- LogP
- -1.5
- TPSA
- 63.3 Ų
- H-Bond Donors
- 2
- H-Bond Acceptors
- 3
- Rotatable Bonds
- 3
- Complexity
- 153
Identifiers (SMILES, InChI)
InChI=1S/C9H11NO2/c10-8(9(11)12)6-7-4-2-1-3-5-7/h1-5,8H,6,10H2,(H,11,12)/t8-/m0/s1
COLNVLDHVKWLRT-QMMMGPOBSA-NSafety Profile
Common Side Effects
- Severe Crashes and Depletion:: Multiple users report horrendous recovery periods with depleted dopamine stores, particularly when combined with alcohol or used for social situations, leading to multi-day crashes
- Caffeine Interaction Changes:: Some report caffeine becomes ineffective or causes unusual reactions when taken with phenylalanine, possibly due to dopamine system saturation
- Overstimulation and Anxiety:: Higher doses or sensitive individuals report jitteriness, loss of inhibition, anxiety, and difficulty sleeping due to extended half-life of metabolites
- Not Universal:: Significant portion of users report no noticeable effects whatsoever despite trying various doses and forms, suggesting strong genetic component to response
References (6)
- [2]Biomedical applications of microbial phenylalanine ammonia lyase: Current status and future prospects
→ Review of PAL enzyme that catabolizes phenylalanine, discussing its therapeutic applications including FDA-approved Palynziq for adult phenylketonuria patients and potential uses in treating tyrosine-related metabolic abnormalities and tumor regression.
- [3]Nutrition in phenylketonuria
→ Comprehensive review of phenylketonuria management through dietary phenylalanine restriction, discussing treatment approaches including medical foods, large neutral amino acids (LNAAs), and enzyme therapy for managing elevated phenylalanine levels.
- [4]How to Optimize the Effectiveness and Safety of Parkinson's Disease Therapy? - A Systematic Review of Drugs Interactions with Food and Dietary Supplements
→ Systematic review identifying food and supplement interactions with Parkinson's medications, noting that phenylalanine and tyrosine from dietary sources can compete with levodopa absorption, affecting treatment efficacy.
- [5]Application of achiral-chiral two-dimensional HPLC for separation of phenylalanine and tryptophan enantiomers in dietary supplement
→ Development of analytical methods to separate and detect phenylalanine enantiomers in dietary supplements, achieving resolution higher than 1.5 for quality control purposes.
- [6]Tyrosine supplementation for phenylketonuria
→ Cochrane review finding weak evidence for tyrosine supplementation in phenylketonuria patients, as tyrosine is the direct metabolite of phenylalanine and may become deficient when phenylalanine is restricted.
- [1]Essential Amino Acids and Protein Synthesis: Insights into Maximizing the Muscle and Whole-Body Response to Feeding
→ Study examining how essential amino acids including phenylalanine from various protein sources influence muscle and whole-body protein synthesis rates, demonstrating the importance of EAA composition in supplements and foods.