Piperine
The primary bioactive alkaloid in black pepper responsible for its pungency, widely used as a bioavailability enhancer that inhibits drug-metabolizing enzymes and intestinal efflux transporters to increase absorption of co-administered compounds.
Piperine is an alkaloid compound extracted from black pepper (Piper nigrum) that acts as a bioavailability enhancer by inhibiting drug-metabolizing enzymes and increasing intestinal permeability. It significantly improves the absorption of various nutrients and pharmaceuticals, most notably increasing curcumin bioavailability by up to 2000%. Piperine is commonly used in supplement formulations to enhance the effectiveness of other compounds rather than as a standalone therapeutic agent.
Overview
Piperine is a nitrogen-containing alkaloid responsible for the pungent taste of black pepper (Piper nigrum) and long pepper (Piper longum). It constitutes 5-9% of black pepper by weight and has been used in Ayurvedic medicine for millennia as a "bioavailability enhancer" — a role now validated by modern pharmacology. Piperine's most clinically significant property is its potent inhibition of hepatic and intestinal drug-metabolizing enzymes, particularly CYP3A4, CYP2D6, CYP1A1, and UDP-glucuronosyltransferases, as well as P-glycoprotein (P-gp) efflux transporters in the intestinal epithelium. By inhibiting these first-pass metabolism and efflux pathways, piperine dramatically increases the systemic bioavailability of co-administered compounds. The most well-documented example is curcumin: piperine increases curcumin bioavailability by approximately 2,000%, transforming an otherwise poorly absorbed polyphenol into a therapeutically relevant oral compound.
Beyond its role as a bioenhancer, piperine possesses intrinsic pharmacological activity. It activates TRPV1 (transient receptor potential vanilloid 1) channels — the same receptors activated by capsaicin — producing thermogenic effects and potentially supporting metabolic rate. Piperine has demonstrated anti-inflammatory activity through inhibition of NF-kB, COX-2, and pro-inflammatory cytokine production, as well as antioxidant effects mediated by upregulation of endogenous antioxidant enzymes. Animal studies have shown antidepressant-like effects, potentially through enhancement of serotonergic and dopaminergic transmission, and neuroprotective properties in models of Alzheimer's and Parkinson's disease. Piperine also stimulates digestive enzyme secretion and enhances pancreatic lipase, amylase, and trypsin activity, supporting its traditional use as a digestive stimulant.
The standardized black pepper extract BioPerine (containing >95% piperine) is the most commonly used commercial form, typically dosed at 5-20 mg alongside target compounds. Piperine enhances the absorption of numerous nutrients and supplements including CoQ10, beta-carotene, selenium, vitamin B6, resveratrol, and various amino acids. However, its enzyme-inhibiting properties warrant caution: piperine can also increase blood levels of prescription medications metabolized by CYP3A4 (including many statins, immunosuppressants, and anticoagulants), creating potential for clinically significant drug interactions. This dual nature — beneficial bioenhancer and potential interaction risk — necessitates careful consideration of concurrent medications.
Mechanism of Action
Piperine (1-piperoylpiperidine) is a pungent alkaloid from Piper nigrum (black pepper) with a multifaceted mechanism of action. Its most clinically significant property is bioavailability enhancement of co-administered drugs and nutrients. Piperine potently inhibits cytochrome P450 enzymes, particularly CYP3A4 and CYP1A2, reducing first-pass hepatic and intestinal metabolism. It also inhibits P-glycoprotein (P-gp/MDR1), an ATP-dependent efflux transporter that actively pumps substrates out of enterocytes back into the intestinal lumen. Additionally, piperine inhibits UDP-glucuronosyltransferase and other phase II conjugation enzymes, further slowing drug metabolism. This triple mechanism of CYP inhibition, P-gp blockade, and phase II enzyme inhibition collectively increases the oral bioavailability of compounds such as curcumin (by up to 2000%), certain beta-lactam antibiotics, and various phytochemicals.
Piperine activates the transient receptor potential vanilloid 1 (TRPV1) channel through a distinct binding mechanism within the vanilloid pocket, differing structurally from capsaicin binding. TRPV1 activation triggers calcium influx, substance P release, and initial nociceptive signaling, accounting for piperine's pungent taste. Repeated exposure leads to TRPV1 desensitization, producing analgesic effects. Piperine also modulates serotonin and endorphin pathways, contributing to its pain-modulating properties.
As an anti-inflammatory agent, piperine suppresses NF-kB nuclear translocation and inhibits MAPK signaling (p38, JNK, ERK), reducing expression of COX-2, iNOS, TNF-alpha, and IL-6. It also demonstrates antioxidant activity by enhancing glutathione levels and superoxide dismutase activity, while exhibiting thermogenic effects through stimulation of catecholamine secretion and metabolic rate enhancement.
Research
Reported Effects
Absorption Synergy:: Most effective when combined with curcumin, with the classic 20mg piperine + 2g curcumin ratio showing ~2000% absorption increase according to users and research. Formulation Quality:: Users emphasize that effectiveness depends heavily on supplement quality, with many products from online retailers containing unreliable or contaminated ingredients. Anti-inflammatory Results:: Users report mixed results for inflammation reduction, with some seeing significant improvements in 3-7 days while others notice no effect even after months. Individual Variability:: Effectiveness appears highly individual, possibly related to genetic factors like HLA-B*35:01 allele which may increase liver toxicity risk
- Most effective when combined with curcumin, with the classic 20mg piperine + 2g curcumin ratio showing ~2000% absorption increase according to users and research
- Users emphasize that effectiveness depends heavily on supplement quality, with many products from online retailers containing unreliable or contaminated ingredients
- Users report mixed results for inflammation reduction, with some seeing significant improvements in 3-7 days while others notice no effect even after months
- Effectiveness appears highly individual, possibly related to genetic factors like HLA-B*35:01 allele which may increase liver toxicity risk
Safety Profile
Piperine is generally safe at culinary doses but may cause gastrointestinal irritation, heartburn, and nausea at higher supplemental doses. It significantly increases the bioavailability of many drugs by inhibiting cytochrome P450 enzymes, which can lead to dangerously elevated blood levels of certain medications. It should be avoided by individuals with gallbladder issues or gastrointestinal ulcers, and caution is advised during pregnancy.
Pharmacokinetic Profile
Piperine — Pharmacokinetic Curve
SubcutaneousQuick Start
- Typical Dose
- 5-20mg piperine taken with other supplements, most commonly 20mg for curcumin bioavailability enhancement
Molecular Structure
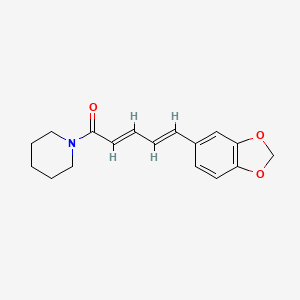
- Formula
- C17H19NO3
- Weight
- 285.34 Da
- PubChem CID
- 638024
- Exact Mass
- 285.1365 Da
- LogP
- 3.5
- TPSA
- 38.8 Ų
- H-Bond Donors
- 0
- H-Bond Acceptors
- 3
- Rotatable Bonds
- 3
- Complexity
- 412
Identifiers (SMILES, InChI)
InChI=1S/C17H19NO3/c19-17(18-10-4-1-5-11-18)7-3-2-6-14-8-9-15-16(12-14)21-13-20-15/h2-3,6-9,12H,1,4-5,10-11,13H2/b6-2+,7-3+
MXXWOMGUGJBKIW-YPCIICBESA-NSafety Profile
Common Side Effects
- Liver Toxicity Risk:: Multiple users report elevated liver enzymes and jaundice from high-potency turmeric-piperine combinations, with at least one reported death
- Drug Metabolism Interference:: Users consistently report that piperine inhibits liver detoxification, potentially causing medications to last longer or accumulate to dangerous levels
- Digestive Discomfort:: Some users experience abdominal pain, though this is often self-limiting and resolved by adjusting dosage or timing
- Iron Depletion:: Several users note that turmeric-piperine combinations can bind iron effectively, potentially leading to anemia in those with low iron status
References (4)
- [1]Pharmacokinetics of piperine after oral administration of Sahastara remedy extract capsules in healthy Thai volunteers
→ Study demonstrated that piperine from traditional remedy extracts achieved peak concentrations of 3.77-6.59 μg/mL with no serious adverse events, supporting its safety profile in healthy volunteers.
- [3]Safety aspects of the use of isolated piperine ingested as a food supplement
→ Safety assessment concluded that isolated piperine used in food supplements at recommended doses appears safe, though potential interactions with medications warrant caution due to enzyme inhibition effects.
- [4]Molecular and pharmacological aspects of piperine as a potential molecule for disease prevention
→ Research demonstrated piperine's pleiotropic activities including antioxidant, anti-inflammatory, and neuroprotective effects through modulation of P-glycoprotein, CYP3A4, and inflammatory cytokines.
- [2]Curcumin-piperine co-supplementation and human health: A comprehensive review
→ Comprehensive review found that piperine co-supplementation provides therapeutic benefits in diabetes, obesity, arthritis, and various cancers by regulating multiple signaling molecules and enhancing drug bioavailability.