Angiotensin-(1-7)
Angiotensin-(1-7) is a bioactive heptapeptide of the renin-angiotensin-aldosterone system (RAAS) that acts as an endogenous ligand for the MAS receptor. It opposes the vasoconstrictive, pro-inflammatory, and pro-fibrotic actions of angiotensin II, exerting vasodilatory, anti-inflammatory, anti-fibrotic, and anti-thrombotic effects with significant implications for cardiovascular, renal, and pulmonary research.
Angiotensin-(1-7) is a seven-amino acid endogenous peptide (Asp-Arg-Val-Tyr-Ile-His-Pro) generated primarily through the enzymatic cleavage of angiotensin II by angiotensin-converting enzyme 2 (ACE2). As the principal effector of the counter-regulatory arm of the renin-angiotensin-aldosterone system (RAAS), Ang-(1-7) opposes the deleterious effects of angiotensin II, making it one of the most intensively studied cardiovascular peptides of the 21st century — particularly since the COVID-19 pandemic revealed the critical importance of the ACE2/Ang-(1-7)/MAS axis.
Overview
Angiotensin-(1-7) occupies a unique position within the RAAS as the primary counter-regulatory peptide to angiotensin II (Ang II). While Ang II — acting through the AT1 receptor — drives vasoconstriction, sodium retention, inflammation, fibrosis, and oxidative stress, Ang-(1-7) — acting through the MAS receptor — promotes vasodilation, natriuresis, anti-inflammation, anti-fibrosis, and anti-oxidative stress responses. This opposing axis creates a dynamic balance that is critical for cardiovascular and renal homeostasis.
The discovery that ACE2 is the primary enzyme responsible for generating Ang-(1-7) from Ang II fundamentally reshaped understanding of the RAAS from a linear cascade into a dual-axis system. When ACE2 activity is reduced — as occurs during SARS-CoV-2 infection, which uses ACE2 as its cell entry receptor — the balance shifts toward Ang II dominance, contributing to the severe cardiovascular, thrombotic, and inflammatory complications observed in COVID-19.
TXA127, a pharmaceutical-grade formulation of Ang-(1-7), has entered clinical trials for multiple indications, including hematopoietic stem cell transplant recovery, pulmonary arterial hypertension, and Duchenne muscular dystrophy, reflecting the broad therapeutic potential of this peptide axis.
Mechanism of Action
Angiotensin-(1-7) exerts its biological effects through several interconnected signaling pathways, primarily via the MAS receptor (MAS1 proto-oncogene receptor):
MAS Receptor Activation: Ang-(1-7) binds to the G protein-coupled receptor MAS, identified by Santos et al. in 2003 as the functional receptor for this peptide. MAS activation triggers phospholipase A2-dependent arachidonic acid release and downstream production of vasodilatory prostanoids, as well as activation of the PI3K/Akt pathway promoting cell survival and anti-apoptotic signaling.
Nitric Oxide (NO) Pathway: Ang-(1-7)/MAS signaling stimulates endothelial nitric oxide synthase (eNOS) phosphorylation via Akt-dependent mechanisms, increasing NO bioavailability. This is a primary mechanism of Ang-(1-7)-mediated vasodilation and endothelial protection, and opposes Ang II-induced NADPH oxidase activation and superoxide generation.
Anti-Inflammatory Signaling: Ang-(1-7) suppresses NF-kB nuclear translocation and reduces expression of pro-inflammatory cytokines including TNF-alpha, IL-6, IL-1beta, and MCP-1. It also inhibits MAPK/ERK signaling pathways that drive inflammatory gene expression downstream of AT1 receptor activation.
Anti-Fibrotic Mechanism: Through MAS receptor activation, Ang-(1-7) inhibits TGF-beta1/Smad signaling, reducing collagen deposition and extracellular matrix accumulation in cardiac, renal, and pulmonary tissues. It also decreases expression of connective tissue growth factor (CTGF) and plasminogen activator inhibitor-1 (PAI-1).
Anti-Thrombotic Activity: Ang-(1-7) stimulates prostacyclin release from endothelial cells and inhibits thromboxane A2 release from platelets. It also reduces tissue factor expression and PAI-1, enhancing fibrinolytic activity. These effects are mediated through both MAS-dependent NO release and direct prostanoid pathway activation.
AT2 Receptor Cross-Talk: Emerging evidence suggests that Ang-(1-7) may also interact with the angiotensin AT2 receptor, which shares protective signaling properties with MAS. Heterodimerization between MAS and AT2 receptors has been demonstrated, potentially amplifying counter-regulatory signaling.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
Research
Cardiovascular Protection
Ang-(1-7) has demonstrated consistent cardioprotective effects across multiple preclinical models. In rodent models of myocardial infarction, Ang-(1-7) infusion reduced infarct size by 25-40%, improved left ventricular function, and attenuated adverse cardiac remodeling. These effects were abolished by the MAS receptor antagonist A-779, confirming MAS-dependent mechanisms. Santos et al. (2004) — Hypertension demonstrated that chronic Ang-(1-7) infusion improved cardiac function in heart failure models. In human studies, plasma Ang-(1-7) levels are reduced in patients with heart failure and correlate inversely with disease severity, suggesting a protective role of this peptide in maintaining cardiac homeostasis.
Renal Protection
The kidney is a major site of Ang-(1-7) production and action. Ang-(1-7) promotes natriuresis and diuresis through actions in the proximal tubule, opposing the sodium-retaining effects of Ang II. In models of diabetic nephropathy, chronic Ang-(1-7) treatment reduced proteinuria, glomerular hypertrophy, and tubulointerstitial fibrosis. Mori et al. (2014) showed that Ang-(1-7) attenuated diabetic kidney injury through inhibition of TGF-beta signaling and oxidative stress. The ACE2/Ang-(1-7)/MAS axis is downregulated in chronic kidney disease, and restoring this axis is a major therapeutic target.
COVID-19 and Acute Lung Injury
The COVID-19 pandemic brought Ang-(1-7) to global attention. SARS-CoV-2 uses ACE2 as its entry receptor, and viral binding triggers ACE2 internalization and downregulation. This reduces Ang-(1-7) production while allowing Ang II to accumulate unopposed, driving the cytokine storm, acute respiratory distress syndrome (ARDS), thrombotic complications, and multi-organ failure characteristic of severe COVID-19. Verdecchia et al. (2020) detailed this mechanistic link. Clinical trials of TXA127 (pharmaceutical Ang-(1-7)) for COVID-19-associated ARDS were initiated, with the rationale that exogenous Ang-(1-7) could restore the protective axis disrupted by viral ACE2 depletion.
Hematopoietic Stem Cell Transplant
TXA127 has been investigated for accelerating hematopoietic recovery after stem cell transplantation. Ang-(1-7) stimulates hematopoietic progenitor cell proliferation through MAS receptor activation on bone marrow stromal cells. A Phase II clinical trial in patients undergoing myeloablative conditioning and allogeneic transplantation demonstrated that TXA127 accelerated neutrophil and platelet recovery. Rodgers et al. (2006) established the hematopoietic effects of Ang-(1-7) in preclinical models.
Cerebrovascular Protection
Ang-(1-7) has demonstrated neuroprotective effects in models of ischemic stroke, reducing infarct volume and improving neurological outcomes. The peptide enhances cerebral blood flow through endothelium-dependent vasodilation and reduces neuroinflammation via suppression of microglial activation. Jiang et al. (2012) showed that intracerebral Ang-(1-7) infusion was protective in a middle cerebral artery occlusion model, with effects blocked by A-779.
Pulmonary Fibrosis
Ang-(1-7) has demonstrated significant anti-fibrotic effects in the lung. In bleomycin-induced pulmonary fibrosis models, Ang-(1-7) reduced collagen deposition, inflammatory cell infiltration, and TGF-beta expression. Meng et al. (2014) showed that Ang-(1-7) attenuated lung fibrosis through inhibition of the MAPK/ERK and Smad2/3 signaling pathways. These findings have relevance not only for idiopathic pulmonary fibrosis but also for post-COVID pulmonary sequelae.
Safety Profile
Angiotensin-(1-7) has demonstrated a favorable safety profile in both preclinical studies and early clinical trials. As an endogenous peptide, it is well-tolerated at physiological and supra-physiological concentrations. In Phase I/II studies of TXA127, the most commonly reported adverse events were mild and transient, including headache, flushing, and dizziness — consistent with its vasodilatory mechanism. Hypotension is a theoretical concern, particularly in patients on concurrent antihypertensive therapy, though clinically significant hypotensive episodes have been uncommon in trials. No serious organ toxicity has been attributed to Ang-(1-7) administration. The peptide does not appear to accumulate with repeated dosing, and its rapid endogenous clearance provides a natural safety margin against overdose effects.
Clinical Research Protocols
- TXA127 (IV infusion): Doses of 100-1000 ng/kg/min have been used in clinical trials for COVID-19 and hematopoietic recovery.
- Subcutaneous injection: Research protocols have used 50-100 mcg/kg/day in preclinical cardiovascular studies.
- Intranasal: Preclinical research has explored intranasal delivery for CNS effects, with doses ranging from 10-100 mcg in rodent models.
- Key trials: TXA127 in hematopoietic stem cell transplant (NCT01397110), TXA127 in COVID-19 ARDS (NCT04375124), TXA127 in Duchenne muscular dystrophy (NCT03177395).
- Duration: Clinical trials have ranged from 7 days (acute COVID-19) to 6 months (muscular dystrophy).
- Routes: Intravenous infusion (most clinical data), subcutaneous injection, intranasal (preclinical).
Subpopulation Research
- Heart failure patients: Reduced circulating Ang-(1-7)/Ang II ratios correlate with heart failure severity (PMID: 15466662). Exogenous Ang-(1-7) improved cardiac output and reduced systemic vascular resistance in preclinical heart failure models.
- Hypertensive populations: ACE2/Ang-(1-7)/MAS axis is suppressed in essential hypertension. Ang-(1-7) infusion acutely reduces blood pressure in hypertensive but not normotensive subjects, suggesting therapeutic specificity.
- Diabetic nephropathy: Ang-(1-7) levels are reduced in diabetic kidney disease. Supplementation attenuated proteinuria and glomerulosclerosis in diabetic animal models (PMID: 24523433).
- COVID-19 patients: Critically ill COVID-19 patients showed markedly reduced ACE2 activity and Ang-(1-7) levels, correlating with worse outcomes (PMID: 32563659).
- Duchenne muscular dystrophy: TXA127 trial in DMD patients based on anti-fibrotic properties; Ang-(1-7) reduced skeletal muscle fibrosis in mdx mice.
- Transplant recipients: TXA127 accelerated hematopoietic recovery after myeloablative conditioning and allogeneic transplant (PMID: 16507768).
- Obese individuals: Visceral adipose tissue has high ACE2 expression; obesity-associated ACE2 dysregulation may contribute to impaired Ang-(1-7) production and metabolic dysfunction.
Pharmacokinetic Profile
Angiotensin-(1-7) — Pharmacokinetic Curve
Subcutaneous injection, Intravenous infusion, Intranasal (research)Ongoing & Future Research
- Oral Ang-(1-7) formulations: Cyclodextrin-based oral formulations are being optimized for chronic administration, potentially enabling long-term cardiovascular and renal protection without injection.
- Long-acting analogues: Modified Ang-(1-7) analogues with enhanced protease resistance and extended half-life are in development. Cyclic analogues and PEGylated forms aim to overcome the extremely short plasma half-life of native Ang-(1-7).
- Post-COVID pulmonary sequelae: Trials investigating Ang-(1-7) for treatment of post-acute sequelae of SARS-CoV-2 (PASC), particularly pulmonary fibrosis and persistent inflammation.
- Cancer: Ang-(1-7) has shown anti-proliferative and anti-angiogenic effects in preclinical cancer models, including lung, prostate, and breast cancer. MAS receptor activation reduces VEGF expression and inhibits tumor angiogenesis.
- Metabolic syndrome: Research is exploring the role of Ang-(1-7) in improving insulin sensitivity, reducing hepatic steatosis, and modulating adipokine secretion in metabolic syndrome.
- Gene therapy approaches: AAV-mediated ACE2 overexpression as a strategy to chronically increase tissue Ang-(1-7) production is being explored in preclinical models of heart failure and kidney disease.
- Biomarker development: The Ang-(1-7)/Ang II ratio is being validated as a prognostic biomarker for cardiovascular risk stratification and COVID-19 severity prediction.
Quick Start
- Route
- Subcutaneous injection, Intravenous infusion, Intranasal (research)
Molecular Structure
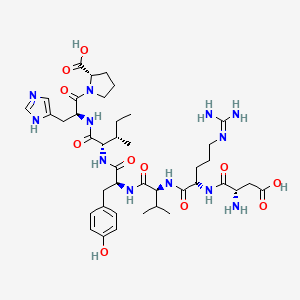
- Formula
- C41H62N12O11
- Weight
- 899.02 Da
- CAS
- 51833-78-4
- PubChem CID
- 123805
- Exact Mass
- 898.4661 Da
- LogP
- -3
- TPSA
- 380 Ų
- H-Bond Donors
- 12
- H-Bond Acceptors
- 14
- Rotatable Bonds
- 25
- Complexity
- 1660
Identifiers (SMILES, InChI)
InChI=1S/C41H62N12O11/c1-5-22(4)33(38(61)50-29(17-24-19-45-20-47-24)39(62)53-15-7-9-30(53)40(63)64)52-36(59)28(16-23-10-12-25(54)13-11-23)49-37(60)32(21(2)3)51-35(58)27(8-6-14-46-41(43)44)48-34(57)26(42)18-31(55)56/h10-13,19-22,26-30,32-33,54H,5-9,14-18,42H2,1-4H3,(H,45,47)(H,48,57)(H,49,60)(H,50,61)(H,51,58)(H,52,59)(H,55,56)(H,63,64)(H4,43,44,46)/t22-,26-,27-,28-,29-,30-,32-,33-/m0/s1
PVHLMTREZMEJCG-GDTLVBQBSA-NResearch Protocols
subcutaneous Injection
- Subcutaneous injection: Research protocols have used 50-100 mcg/kg/day in preclinical cardiovascular studies. - Routes: Intravenous infusion (most clinical data), subcutaneous injection, intranasal (preclinical).
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Subcutaneous injection | 50-100 mcg | Per protocol | —(Route: Subcutaneous Injection) |
intravenous Injection
- Routes: Intravenous infusion (most clinical data), subcutaneous injection, intranasal (preclinical).
intranasal Injection
- Intranasal: Preclinical research has explored intranasal delivery for CNS effects, with doses ranging from 10-100 mcg in rodent models. - Routes: Intravenous infusion (most clinical data), subcutaneous injection, intranasal (preclinical).
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Intranasal | 10-100 mcg | Per protocol | —(Route: Intranasal) |
oral
Ongoing & Future Research - Oral Ang-(1-7) formulations: Cyclodextrin-based oral formulations are being optimized for chronic administration, potentially enabling long-term cardiovascular and renal protection without injection.
Interactions
Peptide Interactions
Both peptides have anti-inflammatory and tissue-protective properties. Potential synergy in wound healing and gastrointestinal protection, though direct combination studies are limited.
Quality Indicators
What to look for
- Human clinical trials conducted
- Well-established safety profile
- Extensive peer-reviewed research base
- Oral administration available
Frequently Asked Questions
References (11)
- [2]Santos, R. A. S. et al Angiotensin-(1-7): an update Regul. Pept. (2004)
- [10]Povlsen, A. L. et al Counter-regulatory renin-angiotensin system in cardiovascular disease Nat. Rev. Cardiol. (2023)
- [8]
- [1]Santos, R. A. S. et al Angiotensin-(1-7) is an endogenous ligand for the G protein-coupled receptor Mas Proc. Natl. Acad. Sci. U.S.A. (2003)
- [3]Verdecchia, P. et al The pivotal link between ACE2 deficiency and SARS-CoV-2 infection Eur. J. Intern. Med. (2020)
- [4]Mori, J. et al Angiotensin 1-7 ameliorates diabetic cardiomyopathy and diastolic dysfunction in db/db mice Mol. Ther. (2014)
- [7]Monteil, V. et al Inhibition of SARS-CoV-2 Infections in Engineered Human Tissues Using Clinical-Grade Soluble Human ACE2 Cell (2020)
- [9]Wagener, G. et al TXA127 (Angiotensin 1-7) for the Treatment of Critically Ill COVID-19 Patients: A Randomized Clinical Trial Crit. Care (2022)
- [11]Magalhaes, G. S. et al Oral formulation of angiotensin-(1-7) attenuates cardiac remodeling and fibrosis in a rat model of heart failure J. Mol. Cell. Cardiol. (2023)
- [5]Rodgers, K. E. et al Effect of angiotensin-(1-7) on hematopoietic recovery after total body irradiation Bone Marrow Transplant. (2006)
- [6]
Angiostatin
Angiostatin is a 38 kDa endogenous angiogenesis inhibitor derived from plasminogen kringle domains 1-4. Discovered by Judah Folkman's laboratory in 1994, it inhibits endothelial cell proliferation and migration by binding cell-surface ATP synthase and inducing apoptosis. Angiostatin demonstrated remarkable tumor dormancy in preclinical models and has been investigated in clinical trials as recombinant human angiostatin for cancer therapy, both as monotherapy and in combination with chemotherapy and radiation.
Angiotensin II
Angiotensin II is a potent vasoactive octapeptide generated by angiotensin-converting enzyme (ACE) from angiotensin I. As the primary effector of the classical renin-angiotensin-aldosterone system (RAAS), it drives vasoconstriction, aldosterone secretion, sympathetic activation, and cellular growth through AT1 receptor signaling, while AT2 receptor activation mediates opposing vasodilatory and anti-proliferative effects.