Endostatin
Endostatin is a 20 kDa C-terminal fragment of collagen XVIII and one of the most potent endogenous angiogenesis inhibitors known. Discovered by O'Reilly and Folkman in 1997, it inhibits endothelial cell proliferation through VEGFR2 inhibition, integrin binding, and HIF-1α suppression. Endostar (recombinant human endostatin) is approved in China for non-small cell lung cancer, while clinical trials in the US and Europe explored its broader oncologic potential.
Endostatin is a 20 kDa proteolytic fragment derived from the C-terminal non-collagenous domain (NC1) of collagen XVIII, identified in 1997 by Michael O'Reilly working in Judah Folkman's laboratory at Children's Hospital Boston. It was the second major endogenous angiogenesis inhibitor discovered by the Folkman group (after angiostatin) and became the most extensively studied member of this class.
Overview
Collagen XVIII is a non-fibrillar collagen of the multiplexin family, located in vascular and epithelial basement membranes. Proteolytic cleavage of its C-terminal NC1 domain by cathepsin L, elastase, and matrix metalloproteinases releases endostatin, which then functions as a potent anti-angiogenic factor. The crystal structure of endostatin reveals a compact globular fold with a zinc-binding site involving histidine residues, and a large basic patch of arginine residues that mediates heparan sulfate proteoglycan binding. Zinc binding is essential for structural integrity and full biological activity.
The discovery of endostatin was driven by the same conceptual framework that produced angiostatin: the hypothesis that tumors produce endogenous inhibitors of angiogenesis that maintain distant metastases in a dormant state. Endostatin was purified from conditioned medium of a murine hemangioendothelioma cell line (EOMA) based on its ability to inhibit endothelial cell proliferation. The subsequent demonstration that recombinant endostatin could cause tumor regression to microscopic dormant nodules — with no acquired drug resistance through multiple treatment cycles — generated extraordinary scientific and public interest.
Mechanism of Action
Endostatin inhibits angiogenesis through multiple parallel mechanisms:
-
VEGFR2 (KDR/Flk-1) Inhibition: Endostatin directly binds VEGFR2 and blocks VEGF-induced receptor phosphorylation and downstream signaling including ERK1/2, p38 MAPK, and Akt pathways. This inhibits VEGF-driven endothelial cell proliferation, survival, and migration. Kim et al. (2002) — J. Biol. Chem.
-
Integrin α5β1 Binding: Endostatin binds integrin α5β1 on endothelial cells, inhibiting integrin-mediated adhesion to fibronectin and disrupting focal adhesion kinase (FAK) and Ras/Raf/ERK signaling. This interaction inhibits endothelial cell migration and survival. Endostatin also interacts with other integrins including αvβ3 and αvβ5. Sudhakar et al. (2003) — Proc. Natl. Acad. Sci. USA
-
HIF-1α Suppression: Endostatin destabilizes hypoxia-inducible factor 1-alpha (HIF-1α) by promoting its degradation through the VHL-proteasome pathway. Since HIF-1α drives transcription of VEGF and other pro-angiogenic genes under hypoxic conditions, its suppression reduces the angiogenic stimulus in the tumor microenvironment. Shin et al. (2008) — Biochem. Biophys. Res. Commun.
-
Heparan Sulfate Proteoglycan Binding: The arginine-rich basic patch of endostatin binds heparan sulfate proteoglycans (HSPGs) including glypicans on the endothelial cell surface. This interaction is required for endostatin's anti-migratory activity and may sequester heparin-binding growth factors (VEGF₁₆₅, FGF-2) in the pericellular space. Karumanchi et al. (2001) — Mol. Cell
-
MMP Inhibition: Endostatin directly inhibits matrix metalloproteinase-2 (MMP-2) catalytic activity and blocks MMP-2 activation by MT1-MMP. Since MMP-2-mediated matrix degradation is essential for endothelial cell invasion during angiogenesis, this provides an additional anti-angiogenic mechanism. Kim et al. (2000) — Cancer Res.
-
Wnt Signaling Modulation: Endostatin modulates the canonical Wnt/β-catenin pathway by interacting with the Wnt co-receptor LRP6, potentially affecting endothelial cell fate decisions during angiogenesis.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
Research
Discovery and Preclinical Efficacy
O'Reilly et al. identified endostatin as a specific inhibitor of endothelial cell proliferation produced by EOMA hemangioendothelioma cells. Recombinant mouse endostatin administered systemically to mice bearing Lewis lung carcinoma, T241 fibrosarcoma, or B16F10 melanoma caused tumor regression to microscopic dormant nodules. Critically, repeated treatment cycles (up to 6 cycles of regression-regrowth-retreatment) did not produce drug resistance, in contrast to conventional chemotherapy. This observation — that targeting genetically stable endothelial cells circumvents tumor-derived drug resistance — was central to Folkman's advocacy for anti-angiogenic therapy. O'Reilly et al. (1997) — Cell
Endostar (rh-Endostatin) in China
Endostar is a recombinant human endostatin with an additional 9-amino acid tag that improves protein stability and extends half-life. It was developed by Simcere Pharmaceutical Group and approved by China's SFDA in 2005 for first-line treatment of NSCLC in combination with cisplatin-based chemotherapy, based on a pivotal phase III trial. In this randomized, double-blind, placebo-controlled trial of 493 patients with stage III/IV NSCLC, Endostar plus vinorelbine/cisplatin (NP) significantly improved overall response rate (35.4% vs. 19.5%, p=0.003) and median time to progression (6.3 vs. 3.6 months) compared to NP alone. Wang et al. (2005) — Zhongguo Fei Ai Za Zhi Subsequent studies have explored Endostar in combination with various chemotherapy regimens for NSCLC, gastric cancer, colorectal cancer, melanoma, breast cancer, and soft tissue sarcoma. Continuous intravenous infusion of Endostar (as pump-delivered infusion over multiple days per cycle) has been investigated as a method to maintain sustained anti-angiogenic plasma levels.
Combination with Radiation Therapy
Endostatin enhances the efficacy of radiation therapy in preclinical models. The proposed mechanism involves vascular normalization — endostatin transiently normalizes the structurally and functionally abnormal tumor vasculature, improving oxygenation and thereby enhancing radiation-induced DNA damage. Timing of endostatin administration relative to radiation is critical, with a "normalization window" during which combination treatment is most effective. Jain (2005) — Science Clinical studies combining Endostar with concurrent chemoradiation in NSCLC have shown promising results, with improved local control rates.
Genomic and Proteomic Effects
Microarray studies revealed that endostatin simultaneously downregulates approximately 12% of the endothelial cell transcriptome, affecting genes involved in proliferation, migration, apoptosis resistance, and vascular remodeling. This broad transcriptomic effect — rather than a single-target mechanism — may explain both the potency and the resistance-free properties of endostatin. Abdollahi et al. (2004) — Mol. Cell The multi-target mechanism of endostatin stands in contrast to single-target agents like bevacizumab (anti-VEGF), potentially explaining why preclinical resistance to endostatin was not observed.
Collagen XVIII Biology and Endostatin Generation
Endogenous endostatin generation occurs through proteolytic processing of collagen XVIII in basement membranes. Circulating endostatin levels are measurable in healthy individuals (at approximately 20-50 ng/mL) and are altered in various disease states. In cancer, elevated circulating endostatin may represent a host anti-angiogenic defense, though the levels are generally insufficient to suppress established tumors. In Down syndrome (trisomy 21), the extra copy of the collagen XVIII gene (COL18A1, located on chromosome 21) results in elevated circulating endostatin levels, which may contribute to the significantly reduced incidence of solid tumors observed in individuals with Down syndrome. Zorick et al. (2001) — Hum. Mol. Genet.
US and European Clinical Trials
In the United States, recombinant human endostatin (rhEndostatin, EntreMed) entered phase I clinical trials in 1999, generating intense media coverage. The phase I study of daily IV bolus rhEndostatin in patients with advanced solid tumors showed safety and tolerability but no objective tumor responses, though minor tumor blood flow reductions were observed by DCE-MRI. Herbst et al. (2002) — J. Clin. Oncol. Subsequent phase II studies of continuous IV infusion rhEndostatin also showed limited single-agent activity. The challenges included: production difficulties (initial problems with protein refolding and lot-to-lot variability), short half-life of unmodified rhEndostatin, and the realization that anti-angiogenic monotherapy was insufficient for advanced cancers. Thomas et al. (2003) — J. Clin. Oncol. Clinical development in the US was ultimately discontinued by EntreMed, while the modified Endostar formulation succeeded in China.
Safety Profile
Endostatin and Endostar have demonstrated favorable safety profiles in clinical studies. In phase I trials of rhEndostatin (EntreMed), the maximum tolerated dose was not reached at doses up to 600 mg/m²/day IV, and no dose-limiting toxicities were identified. The most common adverse effects were mild and included fatigue, injection-site reactions, headache, and gastrointestinal symptoms. Notably, endostatin did not produce the hypertension, proteinuria, bleeding, or thromboembolism associated with VEGF pathway inhibitors, consistent with its distinct multi-target mechanism. Endostar (Chinese formulation) adverse effects in clinical trials and post-marketing surveillance include mild cardiac adverse events (sinus tachycardia, ST-T changes) in approximately 5-10% of patients, which are generally manageable and reversible. Other reported effects include fatigue, mild gastrointestinal symptoms, and rare allergic reactions. Wound healing impairment was not a significant clinical finding, though caution is advised perioperatively. Endostar does not appear to enhance chemotherapy-related myelosuppression when used in combination. Long-term safety data from post-marketing surveillance in China support an acceptable safety profile in combination with standard chemotherapy regimens.
Clinical Research Protocols
- Endostar NSCLC protocol (Chinese pivotal trial): Endostar 7.5 mg/m²/day IV infusion on days 1-14 of each 21-day cycle, in combination with NP (vinorelbine 25 mg/m² days 1, 8 + cisplatin 30 mg/m² days 1-3) for up to 4 cycles. CT assessment every 2 cycles.
- Endostar continuous infusion: Endostar 15 mg/m²/day by continuous IV pump infusion on days 1-7 or 1-14 of 21-day cycles. This approach aims to maintain sustained plasma levels above the anti-angiogenic threshold.
- Phase I US protocol (Herbst et al.): rhEndostatin 15-600 mg/m²/day by IV bolus infusion daily. Dose escalation in cohorts of 3-6 patients. Pharmacokinetic sampling and DCE-MRI tumor perfusion imaging.
- Combination with immunotherapy (investigational): Endostar + PD-1/PD-L1 inhibitor + platinum-based chemotherapy in advanced NSCLC. Monitoring for immune-related and anti-angiogenic adverse events.
- Biomarker assessments: Circulating VEGF, bFGF, and endostatin levels by ELISA. Circulating endothelial cells (CECs) and circulating endothelial progenitor cells (CEPCs) by flow cytometry. DCE-MRI for tumor perfusion.
Pharmacokinetic Profile
Endostatin — Pharmacokinetic Curve
Intravenous infusion (Endostar); subcutaneous (investigational)Quick Start
- Route
- Intravenous infusion (Endostar); subcutaneous (investigational)
Molecular Structure
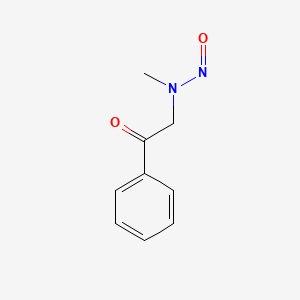
- Formula
- C9H10N2O2
- Weight
- 178.19 Da
- PubChem CID
- 187888
- Exact Mass
- 178.0742 Da
- LogP
- 1.6
- TPSA
- 49.7 Ų
- H-Bond Donors
- 0
- H-Bond Acceptors
- 4
- Rotatable Bonds
- 3
- Complexity
- 188
Identifiers (SMILES, InChI)
InChI=1S/C9H10N2O2/c1-11(10-13)7-9(12)8-5-3-2-4-6-8/h2-6H,7H2,1H3
AAFYOVPTFNNVDN-UHFFFAOYSA-NResearch Protocols
intravenous Injection
Continuous intravenous infusion of Endostar (as pump-delivered infusion over multiple days per cycle) has been investigated as a method to maintain sustained anti-angiogenic plasma levels.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Phase I trials of rhEndostatin | 600 mg | Per protocol | — |
| Combination with NP | 7.5 mg, 25 mg, 30 mg | Per protocol | — |
| General Research Protocol | 15 mg | Per protocol | — |
| General Research Protocol | 15-600 mg | Daily | — |
subcutaneous Injection
Administered via subcutaneous injection.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| General Research Protocol | 600 mg | Per protocol | — |
| Endostar NSCLC protocol (Chinese pivotal trial) | 7.5 mg, 25 mg, 30 mg | Per protocol | — |
| Endostar continuous infusion | 15 mg | Per protocol | — |
| Phase I US protocol (Herbst et al.) | 15-600 mg | Daily | — |
| Biomarker assessments | See literature | Daily | — |
Interactions
Peptide Interactions
Endostatin enhances the efficacy of radiation therapy in preclinical models. The proposed mechanism involves vascular normalization — endostatin transiently normalizes the structurally and functionally abnormal tumor vasculature, improving oxygenation and thereby enhancing radiation-induced DNA d...
What to Expect
What to Expect
Effects begin within hours of administration based on half-life of ~12-13 hours (Endostar, modified form); ~6-8 hours (native rhEndostatin)
Endostar NSCLC protocol (Chinese pivotal trial): Endostar 7.5 mg/m²/day IV infusion on days 1-14 of each 21-day cycle, in combination with NP...
Due to short half-life (~12-13 hours (Endostar, modified form); ~6-8 hours (native rhEndostatin)), effects are expected per-dose; consistent daily...
Endostar NSCLC protocol (Chinese pivotal trial): Endostar 7.5 mg/m²/day IV infusion on days 1-14 of each 21-day cycle, in combination with NP...
Regular administration schedule required; effects are dose-dependent and do not persist between doses
Quality Indicators
What to look for
- Phase 3 clinical trial data available
- Human clinical trials conducted
- Well-established safety profile
- Extensive peer-reviewed research base
Caution
- Short half-life may require frequent dosing
- Injection site reactions reported
Frequently Asked Questions
References (8)
- [6]Wang, J. et al Phase III clinical trial of Endostar combined with NP for advanced NSCLC Zhongguo Fei Ai Za Zhi (2005)
- [1]O'Reilly, M. S. et al Endostatin: an endogenous inhibitor of angiogenesis and tumor growth Cell (1997)
- [2]Herbst, R. S. et al Phase I study of recombinant human endostatin in patients with advanced solid tumors J. Clin. Oncol. (2002)
- [4]Sudhakar, A. et al Human tumstatin and human endostatin exhibit distinct antiangiogenic activities Proc. Natl. Acad. Sci. USA (2003)
- [5]Karumanchi, S. A. et al Cell surface glypicans are low-affinity endostatin receptors Mol. Cell (2001)
- [7]An, J. et al — Endostar combined with chemotherapy for treatment of advanced NSCLC: updated systematic review and meta-analysis Front. Oncol. (2021)
- [3]
- [8]
Endomorphin-2
Endomorphin-2 (Tyr-Pro-Phe-Phe-NH₂) is an endogenous opioid tetrapeptide with high mu-opioid receptor selectivity, discovered alongside endomorphin-1. It differs by a single residue (Phe³ vs Trp³) and shows a distinct distribution pattern with predominant spinal cord localization, suggesting complementary roles in supraspinal (EM-1) versus spinal (EM-2) analgesia.
Enfuvirtide
Enfuvirtide (T-20, Fuzeon) is a 36-amino acid synthetic peptide derived from the HR2 region of HIV-1 gp41. It is the first FDA-approved HIV fusion inhibitor (2003), blocking viral entry by preventing six-helix bundle formation required for membrane fusion.