Davunetide (AL-108)
Davunetide (AL-108) is an intranasal formulation of the NAP peptide (NAPVSIPQ) derived from activity-dependent neuroprotective protein (ADNP), developed by Allon Therapeutics for clinical trials in schizophrenia cognitive impairment, progressive supranuclear palsy, and mild cognitive impairment.
Davunetide (AL-108) is the clinical-stage intranasal formulation of the NAP peptide (NAPVSIPQ), developed by Allon Therapeutics Inc. (Vancouver, Canada) for neurodegenerative and neuropsychiatric indications. It is the most extensively clinically tested formulation of the ADNP-derived neuroprotective peptide discovered by Illana Gozes at Tel Aviv University.
Overview
Davunetide represents the translational bridge between NAP peptide basic research and clinical application. Allon Therapeutics developed the intranasal formulation to exploit NAP's ability to cross the blood-brain barrier via olfactory and trigeminal nerve pathways, achieving therapeutic CNS concentrations without systemic injection. The drug demonstrated neuroprotective and procognitive effects in multiple Phase 2 trials, including statistically significant improvements in memory performance in patients with amnestic MCI and enhanced cognitive function in schizophrenia patients. However, the pivotal Phase 2/3 trial in progressive supranuclear palsy (a pure tauopathy) failed to meet primary endpoints, leading to discontinuation of Allon Therapeutics' clinical program. Despite this setback, the mechanistic rationale and Phase 2 efficacy signals continue to generate research interest.
Mechanism of Action
Davunetide shares the mechanism of the NAP peptide: it binds to end-binding proteins EB1 and EB3 at microtubule plus-ends via its SxIP motif, stabilizing microtubule dynamics and promoting proper cytoskeletal function. This microtubule stabilization protects against tau-mediated neurodegeneration by compensating for the loss of tau's normal microtubule-stabilizing function when tau becomes hyperphosphorylated and detaches. Additionally, davunetide reduces tau hyperphosphorylation, promotes autophagic clearance of tau aggregates, and protects neurons from oxidative stress and excitotoxicity.
The intranasal formulation leverages the direct nose-to-brain transport pathway. After nasal deposition, the peptide is absorbed through the olfactory epithelium and transported via olfactory nerve axons directly to the olfactory bulb and subsequently to the hippocampus, cortex, and other brain regions -- bypassing the blood-brain barrier entirely. This route achieves higher CNS bioavailability than systemic administration for this peptide class.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
Research
Continued Development: CP201
Following the Allon Therapeutics discontinuation, the ADNP/NAP technology was licensed to Coronis Neurosciences (later renamed to Shire and subsequently acquired). The compound was redesignated CP201 for potential development in ADNP syndrome (Helsmoortel-Van der Werf syndrome), where the mechanistic rationale -- replacing deficient ADNP function -- is more direct than in sporadic tauopathies.
TURNS Trial: Schizophrenia Cognition
The Treatment Units for Research on Neurocognition and Schizophrenia (TURNS) program evaluated davunetide for cognitive impairment associated with schizophrenia (CIAS). Javitt et al. reported a Phase 2 trial in 63 schizophrenia patients receiving davunetide 30 mg/day intranasal or placebo for 12 weeks. Davunetide-treated patients showed improvements on the MATRICS Consensus Cognitive Battery (MCCB) composite score, with the most robust effects on attention/vigilance and working memory domains. Daily functioning measures (UCSD Performance-based Skills Assessment, UPSA) also showed improvement. Javitt D.C. et al. (2012) -- Schizophr. Res.
Phase 2/3 Trial in Progressive Supranuclear Palsy
Boxer et al. conducted the pivotal Phase 2/3 trial of davunetide in 313 patients with probable or possible PSP -- a pure tauopathy without significant amyloid pathology. Patients received davunetide 30 mg/day intranasal or placebo for 52 weeks. The trial failed to meet its co-primary endpoints: no significant differences were observed in the PSP Rating Scale (PSPRS) or the Schwab and England Activities of Daily Living Scale (SEADL). Secondary endpoints (cognitive tests, brain atrophy by MRI, CSF biomarkers) also showed no significant treatment effects. This negative result led to discontinuation of Allon Therapeutics' davunetide development program. Boxer A.L. et al. (2014) -- Lancet Neurol.
Post-Hoc Analyses and Lessons Learned
Morimoto et al. conducted post-hoc analyses of the PSP trial data, examining whether davunetide might have benefited specific patient subgroups. Several hypotheses have been proposed for the PSP trial failure: (1) PSP involves 4R tau isoforms, while NAP's preclinical efficacy was primarily demonstrated in 3R or mixed tau models; (2) the advanced stage of PSP at enrollment may have been beyond the point of rescue by microtubule stabilization; (3) intranasal delivery may not have achieved sufficient brainstem/basal ganglia concentrations relevant to PSP pathology; (4) the dose may have been suboptimal given the non-linear dose-response observed in the MCI trial. Morimoto B.H. et al. (2013) -- J. Alzheimers Dis.
Phase 2 Trial in Amnestic Mild Cognitive Impairment
Javitt et al. conducted a randomized, double-blind, placebo-controlled Phase 2 trial of davunetide in 144 patients with amnestic MCI (a prodromal stage of Alzheimer's disease). Patients received intranasal davunetide at 5 mg/day or 30 mg/day versus placebo for 12 weeks. The 5 mg dose group showed statistically significant improvements on the Paired Associates Learning (PAL) test -- a measure of visual episodic memory known to decline early in AD -- compared to placebo. The 30 mg dose showed trends toward improvement but did not reach statistical significance, suggesting a possible U-shaped dose-response curve. Javitt D.C. et al. (2012) -- Schizophr. Res.
Safety Profile
Davunetide demonstrated a favorable safety profile across all clinical trials. In the 12-week MCI and schizophrenia trials (n=207 combined davunetide-treated patients), adverse events were comparable to placebo. The most common side effects were mild nasal irritation (12% vs 8% placebo), headache (8% vs 6%), and rhinorrhea (5% vs 3%). In the 52-week PSP trial (n=157 davunetide-treated), the safety profile remained consistent with shorter trials. No serious adverse events were attributed to davunetide. No evidence of systemic immunogenicity, organ toxicity, or hematological abnormalities was observed. The intranasal route avoided gastrointestinal side effects and first-pass hepatic metabolism. Davunetide did not interfere with concomitant medications, including antipsychotics in schizophrenia trials.
Pharmacokinetic Profile
Davunetide (AL-108) — Pharmacokinetic Curve
Intranasal sprayQuick Start
- Route
- Intranasal spray
Molecular Structure
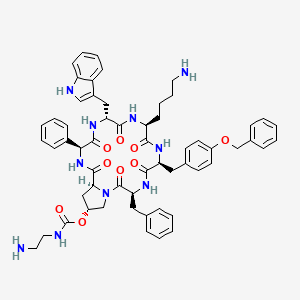
- Formula
- C36H64N10O12
- Weight
- 824.97 Da
- CAS
- 329218-17-1
- PubChem CID
- 9941444
- Exact Mass
- 1046.5014 Da
- LogP
- 4.7
- TPSA
- 281 Ų
- H-Bond Donors
- 9
- H-Bond Acceptors
- 11
- Rotatable Bonds
- 18
- Complexity
- 1940
Identifiers (SMILES, InChI)
InChI=1S/C58H66N10O9/c59-27-13-12-22-46-52(69)64-47(30-38-23-25-42(26-24-38)76-36-39-16-6-2-7-17-39)53(70)66-49(31-37-14-4-1-5-15-37)57(74)68-35-43(77-58(75)61-29-28-60)33-50(68)55(72)67-51(40-18-8-3-9-19-40)56(73)65-48(54(71)63-46)32-41-34-62-45-21-11-10-20-44(41)45/h1-11,14-21,23-26,34,43,46-51,62H,12-13,22,27-33,35-36,59-60H2,(H,61,75)(H,63,71)(H,64,69)(H,65,73)(H,66,70)(H,67,72)/t43-,46+,47+,48-,49+,50+,51+/m1/s1
VMZMNAABQBOLAK-DBILLSOUSA-NResearch Protocols
intranasal Injection
Davunetide (AL-108) is the clinical-stage intranasal formulation of the NAP peptide (NAPVSIPQ), developed by Allon Therapeutics Inc. Allon Therapeutics developed the intranasal formulation to exploit NAP's ability to cross the blood-brain barrier via olfactory and trigeminal nerve pathways, achievin
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| General Research Protocol | 5 mg, 30 mg | Twice daily | 12 weeks(Route: Intranasal) |
Interactions
Peptide Interactions
Microtubule stabilization (davunetide) plus neurotrophic support via BDNF/NGF upregulation (Semax). Non-overlapping mechanisms for comprehensive neuroprotection.
Microtubule stabilization (davunetide) plus neurotrophic support via BDNF/NGF upregulation (Semax). Non-overlapping mechanisms for comprehensive neuroprotection.
What to Expect
What to Expect
Rapid onset expected; half-life of ~30 minutes (intranasal) indicates fast-acting pharmacokinetics
Patients received intranasal davunetide at 5 mg/day or 30 mg/day versus placebo for 12 weeks.
Patients received davunetide 30 mg/day intranasal or placebo for 52 weeks.
Continued use as directed
Quality Indicators
What to look for
- Human clinical trials conducted
- Well-established safety profile
Red flags
- Significant side effect risk noted
Frequently Asked Questions
References (7)
- [7]Gozes I -- ADNP/NAP: Current Evidence and Future Perspectives for Autism and Beyond Front. Endocrinol. (2023)
- [8]Grigg I. et al -- Tauopathy mechanisms and therapeutic strategies: Implications for Alzheimer's disease and progressive supranuclear palsy Front. Neurol. (2022)
- [1]Javitt, D.C. et al Effect of the neuroprotective peptide davunetide (AL-108) on cognition and functional capacity in schizophrenia Schizophr. Res. (2012)
- [2]Boxer, A.L. et al Davunetide in patients with progressive supranuclear palsy: a randomised, double-blind, placebo-controlled phase 2/3 trial Lancet Neurol. (2014)
- [3]Morimoto, B.H. et al Davunetide: a review of safety and efficacy data with a focus on neurodegenerative diseases Expert Rev. Clin. Pharmacol. (2013)
- [4]Gozes, I. et al NAP: Research and development of a peptide derived from activity-dependent neuroprotective protein (ADNP) CNS Drug Rev. (2005)
- [6]
Danuglipron
Danuglipron (PF-06882961) is an oral non-peptide small molecule GLP-1 receptor agonist developed by Pfizer. Originally investigated as a twice-daily formulation, a modified-release once-daily version is in development for type 2 diabetes and obesity.
Decapeptide-12
Decapeptide-12 is a synthetic oligopeptide that potently inhibits tyrosinase activity, reducing melanin production in melanocyte cultures. It is under investigation as a topical agent for hyperpigmentation and skin-lightening applications in animal models.