NAP Peptide (NAPVSIPQ)
NAP (NAPVSIPQ) is an eight-amino acid peptide derived from activity-dependent neuroprotective protein (ADNP) that provides femtomolar-level neuroprotection through microtubule stabilization and tau interaction, with research in Alzheimer's disease, autism spectrum disorder, and neurodegeneration.
NAP (sequence: NAPVSIPQ, Asn-Ala-Pro-Val-Ser-Ile-Pro-Gln) is an octapeptide representing the smallest active fragment of activity-dependent neuroprotective protein (ADNP), a critical homeostatic regulator of brain development and function. Discovered by Professor Illana Gozes at Tel Aviv University, NAP provides neuroprotection at femtomolar concentrations (10^-15 M) -- among the most potent neuroprotective activities ever documented for a peptide.
Overview
ADNP is one of the most essential proteins for brain development -- ADNP knockout is embryonically lethal in mice, with severe neural tube closure defects. ADNP contains an SH3 (Src homology 3) binding domain, and the NAP octapeptide maps to this domain. The NAP motif mediates ADNP's interaction with microtubules through the SxIP motif (Ser-x-Ile-Pro), which binds end-binding proteins (EB1 and EB3) at microtubule plus-ends. This interaction stabilizes the dynamic instability of microtubules, protecting them from disassembly and promoting proper axonal transport.
Mutations in the ADNP gene cause Helsmoortel-Van der Werf syndrome, a form of autism spectrum disorder characterized by intellectual disability, motor delays, and facial dysmorphism. NAP peptide has shown corrective effects in cellular and animal models of this syndrome, restoring microtubule dynamics and improving behavioral outcomes. Beyond autism, NAP protects against a broad spectrum of neurotoxic insults including amyloid-beta toxicity, oxidative stress, excitotoxicity, and tau hyperphosphorylation.
Mechanism of Action
NAP exerts neuroprotection through a primary mechanism centered on microtubule stabilization via the SxIP motif. The Ser-Ile-Pro sequence within NAP binds to end-binding proteins EB1 and EB3, which are master regulators of microtubule plus-end dynamics. By enhancing EB1/EB3 binding at microtubule tips, NAP promotes microtubule polymerization, reduces catastrophic disassembly events, and maintains the cytoskeletal architecture essential for axonal transport, dendritic morphology, and synaptic function.
NAP also interacts directly with tau protein. Under normal conditions, tau stabilizes microtubules by binding along their surface. In tauopathies, hyperphosphorylated tau dissociates from microtubules, aggregates into paired helical filaments and neurofibrillary tangles, and simultaneously destabilizes the microtubule network. NAP compensates for tau loss-of-function by providing an alternative mechanism of microtubule stabilization, effectively bypassing the requirement for normal tau function. Additionally, NAP has been shown to reduce tau hyperphosphorylation and promote clearance of pathological tau species through autophagy-related mechanisms.
Secondary protective mechanisms include inhibition of oxidative stress through upregulation of antioxidant enzymes, prevention of apoptosis via Bcl-2 family regulation, and modulation of calcium homeostasis.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
Research
Microtubule Dynamics and EB1/EB3 Interaction
Oz et al. identified the SxIP motif within NAP as the molecular basis for its microtubule interaction. NAP binds EB1 and EB3 at microtubule plus-ends, promoting polymerization and reducing catastrophe frequency. This interaction is critical for maintaining axonal transport of mitochondria, synaptic vesicles, and neurotrophic factor receptors. Disruption of microtubule dynamics is a common pathological feature across neurodegenerative diseases, making NAP's mechanism broadly relevant. Oz S. et al. (2014) -- J. Biol. Chem.
Tau Pathology Protection
Vulih-Shultzman et al. showed that NAP protects against tau hyperphosphorylation and aggregation in multiple model systems. In zinc-induced tau toxicity models, NAP prevented tau-mediated microtubule destruction and maintained neuronal viability. NAP treatment also reduced levels of insoluble tau aggregates in the hippocampus of tau-transgenic mice, suggesting direct interference with the tau aggregation cascade. Vulih-Shultzman I. et al. (2007) -- J. Pharmacol. Exp. Ther.
ADNP Syndrome / Helsmoortel-Van der Werf Syndrome
Mutations in the ADNP gene account for approximately 0.17% of autism spectrum disorder cases globally. Hacohen-Kleiman et al. demonstrated that NAP peptide corrects microtubule deficits in ADNP-mutant neurons, restoring dendritic spine morphology and synaptic function. In ADNP-heterozygous mice (modeling the human haploinsufficiency), intranasal NAP improved social behavior, object recognition memory, and reduced repetitive behaviors. Hacohen-Kleiman G. et al. (2018) -- Mol. Psychiatry
Fetal Alcohol Syndrome Protection
NAP protects developing neurons from ethanol toxicity, preventing fetal alcohol spectrum disorder (FASD)-related damage in animal models. Pregnant mice exposed to alcohol and co-treated with NAP produced offspring with significantly fewer learning deficits and reduced neuronal apoptosis compared to alcohol-only controls. This protection is mediated through microtubule stabilization in developing neurons, which are particularly vulnerable to ethanol-induced cytoskeletal disruption. Spong C. Y. et al. (2001) -- J. Pharmacol. Exp. Ther.
Femtomolar Neuroprotection
The defining characteristic of NAP is its extraordinary potency. Bassan et al. demonstrated that NAP protects neurons from diverse toxic insults -- including electrical blockade (tetrodotoxin), beta-amyloid peptide, NMDA excitotoxicity, and oxidative stress (hydrogen peroxide) -- at concentrations as low as 10^-15 M (femtomolar). This represents one of the most potent neuroprotective activities documented for any compound and suggests a catalytic or amplification mechanism rather than simple receptor occupancy. Bassan M. et al. (1999) -- J. Neurochem.
Safety Profile
NAP has demonstrated an excellent safety profile across multiple preclinical studies and limited clinical testing (as davunetide/AL-108). In animal studies, chronic intranasal NAP administration showed no significant toxicity, behavioral abnormalities, or organ pathology. As an endogenous-derived peptide fragment, NAP operates within established physiological pathways. Clinical trials of the davunetide formulation in humans showed good tolerability with mild adverse events limited to nasal irritation and headache at therapeutic doses. The peptide does not appear to affect peripheral microtubule function at neuroprotective concentrations, likely because its femtomolar potency in neurons reflects CNS-specific amplification mechanisms involving ADNP-related signaling.
Pharmacokinetic Profile
NAP Peptide (NAPVSIPQ) — Pharmacokinetic Curve
Intranasal, Intravenous, Intracerebroventricular (research)Quick Start
- Route
- Intranasal, Intravenous, Intracerebroventricular (research)
Molecular Structure
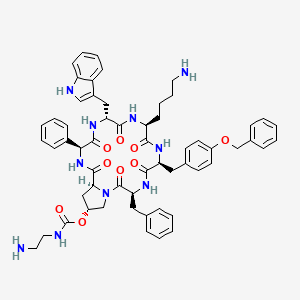
- Formula
- C36H64N10O12
- Weight
- 824.97 Da
- CAS
- 329218-17-1
- PubChem CID
- 9941444
- Exact Mass
- 1046.5014 Da
- LogP
- 4.7
- TPSA
- 281 Ų
- H-Bond Donors
- 9
- H-Bond Acceptors
- 11
- Rotatable Bonds
- 18
- Complexity
- 1940
Identifiers (SMILES, InChI)
InChI=1S/C58H66N10O9/c59-27-13-12-22-46-52(69)64-47(30-38-23-25-42(26-24-38)76-36-39-16-6-2-7-17-39)53(70)66-49(31-37-14-4-1-5-15-37)57(74)68-35-43(77-58(75)61-29-28-60)33-50(68)55(72)67-51(40-18-8-3-9-19-40)56(73)65-48(54(71)63-46)32-41-34-62-45-21-11-10-20-44(41)45/h1-11,14-21,23-26,34,43,46-51,62H,12-13,22,27-33,35-36,59-60H2,(H,61,75)(H,63,71)(H,64,69)(H,65,73)(H,66,70)(H,67,72)/t43-,46+,47+,48-,49+,50+,51+/m1/s1
VMZMNAABQBOLAK-DBILLSOUSA-NResearch Protocols
intracerebroventricular Injection
Administered via intracerebroventricular.
intranasal Injection
In ADNP-heterozygous mice (modeling the human haploinsufficiency), intranasal NAP improved social behavior, object recognition memory, and reduced repetitive behaviors. In animal studies, chronic intranasal NAP administration showed no significant toxicity, behavioral abnormalities, or organ patholo
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| General Research Protocol | 5-30 mg | Daily | 2-8 weeks(Route: Intranasal) |
intravenous Injection
Administered via intravenous injection.
Interactions
Peptide Interactions
- NAP stabilizes microtubules and protects against tauopathy while Semax upregulates BDNF/NGF for neurotrophic support. Different mechanisms (cytoskeletal protection vs. neurotrophic signaling) suggest additive neuroprotection without receptor competition.
NAP stabilizes microtubules and protects against tauopathy while Semax upregulates BDNF/NGF for neurotrophic support. Different mechanisms (cytoskeletal protection vs. neurotrophic signaling) suggest additive neuroprotection without receptor competition.
NAP protects the microtubule network required for mitochondrial transport while SS-31 protects mitochondrial membrane integrity. Combined, they could maintain both the transport system and the organelles it carries.
NAP provides microtubule-based neuroprotection while humanin blocks apoptosis through Bax/Bid sequestration. Complementary protection: cytoskeletal integrity (NAP) plus anti-apoptotic signaling (humanin).
What to Expect
What to Expect
Rapid onset expected; half-life of ~30 minutes (intranasal) indicates fast-acting pharmacokinetics
Due to short half-life (~30 minutes (intranasal)), effects are expected per-dose; consistent daily administration maintains therapeutic levels
Regular administration schedule required; effects are dose-dependent and do not persist between doses
Quality Indicators
What to look for
- Human clinical trials conducted
- Multiple peer-reviewed studies available
Frequently Asked Questions
References (10)
- [8]Gozes I -- ADNP/NAP: Current Evidence and Future Perspectives for Autism and Beyond Front. Endocrinol. (2023)
- [9]Hacohen-Kleiman G. et al -- NAP (davunetide) rescues dendritic spine pathology and social behavior in ADNP-deficient mice Biol. Psychiatry. (2023)
- [10]Ivashko-Pachima Y. et al -- NAP protects against tau hyperphosphorylation through SxIP-mediated microtubule interaction J. Mol. Neurosci. (2022)
- [6]Spong, C. Y. et al Prevention of fetal demise and growth restriction in a mouse model of fetal alcohol syndrome J. Pharmacol. Exp. Ther. (2001)
- [1]Bassan, M. et al Complete sequence of a novel protein containing a femtomolar-activity-dependent neuroprotective peptide J. Neurochem. (1999)
- [2]Gozes, I. et al NAP: Research and development of a peptide derived from activity-dependent neuroprotective protein (ADNP) CNS Drug Rev. (2005)
- [4]Oz, S. et al The NAP motif of activity-dependent neuroprotective protein (ADNP) regulates dendritic spines through microtubule end binding proteins Mol. Psychiatry (2014)
- [7]Gozes, I Activity-dependent neuroprotective protein: From gene to drug candidate Pharmacol. Ther. (2007)
- [3]Vulih-Shultzman, I. et al Activity-dependent neuroprotective protein snippet NAP reduces tau hyperphosphorylation and enhances learning in a novel transgenic mouse model J. Pharmacol. Exp. Ther. (2007)
- [5]Hacohen-Kleiman, G. et al ADNP: Providing neuroprotection in autism — the missing link Mol. Psychiatry (2018)
Nafarelin
Nafarelin is a synthetic GnRH agonist with a D-Nal(2)6 substitution, approximately 200x more potent than native GnRH. Marketed exclusively as an intranasal spray (Synarel, Pfizer), it is FDA-approved for endometriosis and central precocious puberty, and widely used for IVF long-protocol downregulation.
Neuropeptide Y (NPY)
Neuropeptide Y (NPY) is a 36-amino acid peptide and the most abundant neuropeptide in the mammalian brain, playing critical roles in appetite regulation, anxiety modulation, stress resilience, cardiovascular control, and bone metabolism through Y1–Y5 receptor subtypes.