Triptorelin
Triptorelin is a synthetic GnRH agonist (D-Trp6-GnRH) used clinically for prostate cancer, endometriosis, precocious puberty, and uterine fibroids. Available as 1-, 3-, and 6-month depot formulations, it achieves sustained gonadotropin suppression through GnRH receptor desensitization after an initial hormonal flare.
Triptorelin (D-Trp6-GnRH) is a synthetic decapeptide analog of gonadotropin-releasing hormone (GnRH) in which the native glycine at position 6 is replaced with D-tryptophan. This single substitution confers resistance to enzymatic degradation, dramatically extending the half-life from minutes to hours and increasing receptor binding affinity approximately 100-fold compared to native GnRH.
Overview
Triptorelin was developed as part of the systematic effort by Andrew Schally's laboratory to create GnRH superagonists with enhanced pharmacological profiles. The D-tryptophan substitution at position 6 was selected from a series of D-amino acid replacements because it provided optimal receptor binding affinity and metabolic stability. Triptorelin is distinguished from other GnRH agonists (leuprolide, goserelin, buserelin) primarily by its formulation options and regional prescribing patterns — it is particularly widely used in Europe and has gained notable attention in the bodybuilding and anabolic-androgenic steroid (AAS) recovery community for HPTA restart protocols.
Mechanism of Action
Triptorelin binds the GnRH receptor (GnRHR) on anterior pituitary gonadotroph cells with approximately 100-fold greater affinity than native GnRH. The D-Trp6 substitution prevents cleavage by endopeptidases at the Gly6-Leu7 bond (the primary degradation site of native GnRH), resulting in prolonged receptor occupancy. Upon initial administration, triptorelin causes a surge in LH and FSH release (the "flare" response), which elevates testosterone in men and estradiol in women for approximately 7-14 days. With continued receptor stimulation from depot formulations, gonadotroph GnRHR undergoes internalization and transcriptional downregulation. By weeks 2-4, LH and FSH are suppressed to castrate levels, and downstream sex steroid production falls correspondingly. Testosterone reaches castrate levels (<50 ng/dL) in >96% of men by week 4.
Research
Endometriosis
Triptorelin depot creates a hypoestrogenic state that induces atrophy of ectopic endometrial tissue. Prentice et al. (2000) reviewed GnRH agonist efficacy in endometriosis, confirming symptom relief comparable to danazol with a more favorable side effect profile. Treatment duration is typically limited to 6 months unless add-back therapy is used.
Central Precocious Puberty
Triptorelin 3.75 mg monthly or 11.25 mg every 3 months effectively suppresses pubertal progression in children with central precocious puberty. Carel et al. (2004) demonstrated preservation of adult height potential with long-term treatment and normal pubertal resumption upon discontinuation.
HPTA Restart and PCT Microdosing
In the anabolic steroid recovery community, a single low-dose triptorelin injection (typically 100 mcg SC) has been proposed for hypothalamic-pituitary-testicular axis (HPTA) restart after prolonged AAS use. The rationale, described by Tan & Scally, is that a single sub-therapeutic dose stimulates a brief gonadotropin surge (exploiting the flare effect) without causing sustained downregulation. This approach remains controversial and lacks rigorous controlled trial data; inappropriate dosing risks prolonged suppression rather than recovery.
Prostate Cancer
Triptorelin is approved for androgen deprivation therapy (ADT) in advanced prostate cancer. Heyns et al. (2003) demonstrated non-inferiority of triptorelin 3-month depot versus leuprolide in achieving and maintaining castrate testosterone levels. The SPCG-7/SFUO-3 trial showed that adding local radiotherapy to triptorelin-based ADT significantly improved prostate cancer-specific survival in locally advanced disease Widmark et al. (2009).
Safety Profile
Triptorelin has an extensive clinical safety record spanning decades. Adverse effects are primarily related to the intended hypogonadal state: hot flashes (50-80%), decreased libido, erectile dysfunction or vaginal dryness, fatigue, and mood disturbances. The initial testosterone/estradiol flare (days 1-14) can transiently worsen disease — bone pain, urinary obstruction, or spinal cord compression in prostate cancer patients — necessitating anti-androgen cover (bicalutamide 50 mg daily for 2-4 weeks). Long-term ADT carries risks of bone mineral density loss (2-5% annually), metabolic syndrome, increased cardiovascular risk, and sarcopenia. Injection site reactions (pain, erythema, induration) occur in approximately 5% of patients. All hormonal effects are reversible upon discontinuation, with testosterone recovery typically within 2-4 months after the last depot.
Pharmacokinetic Profile
Triptorelin — Pharmacokinetic Curve
Intramuscular depot injectionQuick Start
- Route
- Intramuscular depot injection
Molecular Structure
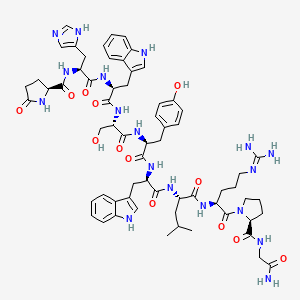
- Formula
- C64H82N18O13
- Weight
- 1311.5 Da
- CAS
- 57773-63-4
- PubChem CID
- 25074470
- Exact Mass
- 1310.6309 Da
- LogP
- -0.3
- TPSA
- 490 Ų
- H-Bond Donors
- 17
- H-Bond Acceptors
- 15
- Rotatable Bonds
- 33
- Complexity
- 2710
Identifiers (SMILES, InChI)
InChI=1S/C64H82N18O13/c1-34(2)23-46(56(88)75-45(13-7-21-69-64(66)67)63(95)82-22-8-14-52(82)62(94)72-31-53(65)85)76-58(90)48(25-36-28-70-42-11-5-3-9-40(36)42)78-57(89)47(24-35-15-17-39(84)18-16-35)77-61(93)51(32-83)81-59(91)49(26-37-29-71-43-12-6-4-10-41(37)43)79-60(92)50(27-38-30-68-33-73-38)80-55(87)44-19-20-54(86)74-44/h3-6,9-12,15-18,28-30,33-34,44-52,70-71,83-84H,7-8,13-14,19-27,31-32H2,1-2H3,(H2,65,85)(H,68,73)(H,72,94)(H,74,86)(H,75,88)(H,76,90)(H,77,93)(H,78,89)(H,79,92)(H,80,87)(H,81,91)(H4,66,67,69)/t44-,45-,46-,47-,48+,49-,50-,51-,52-/m0/s1
VXKHXGOKWPXYNA-PGBVPBMZSA-NResearch Protocols
intramuscular Injection
Administered via intramuscular injection.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| General Research Protocol | 50 mg, 3.75 mg, 11.25 mg, 22.5 mg, 100 mcg | Daily | 2-4 weeks |
Interactions
Peptide Interactions
Combining triptorelin with bicalutamide (50-150 mg daily) or enzalutamide blocks both testicular (via gonadotropin suppression) and adrenal androgen pathways. This provides more complete androgen receptor suppression than either agent alone and prevents flare-related disease progression during th...
What to Expect
What to Expect
Effects begin within hours of administration based on half-life of 7.6 hours (free peptide); depot-dependent effective duration
Anti-androgen flare protection with bicalutamide 50 mg daily starting 7 days before the first injection and continuing for 2-4 weeks.
The initial testosterone/estradiol flare (days 1-14) can transiently worsen disease — bone pain, urinary obstruction, or spinal cord compression in...
Testosterone reaches castrate levels (<50 ng/dL) in >96% of men by week 4.
Hormonal recovery after discontinuation of monthly depot occurs within 2-3 months; 3-month and 6-month depots may require 3-6 months for full...
Quality Indicators
What to look for
- Multiple peer-reviewed studies available
Caution
- Injection site reactions reported
Frequently Asked Questions
References (10)
- [9]Neven et al — Long-term outcomes of GnRH agonist treatment in central precocious puberty Hum. Reprod. Update (2022)
- [8]Klotz et al — Degarelix vs triptorelin in prostate cancer: cardiovascular safety analysis Eur. Urol. (2023)
- [10]Boehm et al — GnRH agonist depot formulations: pharmacokinetic advances and clinical implications J. Clin. Endocrinol. Metab. (2024)
- [1]
- [2]
- [3]
- [7]
- [6]Prentice A et al Gonadotrophin-releasing hormone analogues for endometriosis Cochrane Database Syst Rev (2000)
- [4]Widmark A et al Endocrine treatment, with or without radiotherapy, in locally advanced prostate cancer (SPCG-7) Lancet (2009)
- [5]Carel JC et al Treatment of central precocious puberty by GnRH agonists Ann Endocrinol (Paris) (2004)
Tripeptide-29
Tripeptide-29 is a collagen-fragment tripeptide and GHK analogue that stimulates procollagen I synthesis and promotes wound healing by mimicking a type I collagen signaling fragment to activate dermal fibroblasts.
TSH (Thyrotropin)
Thyroid-stimulating hormone (TSH, thyrotropin) is a 211 amino acid glycoprotein hormone secreted by the anterior pituitary that drives thyroid hormone synthesis. Recombinant TSH (thyrotropin alfa) is used in thyroid cancer management and diagnostic evaluation, with peptide analogs under investigation.