MOTS-c
MOTS-c is a mitochondrial-derived peptide composed of 16 amino acids that regulates cellular energy metabolism, promotes metabolic homeostasis, and has demonstrated effects on insulin sensitivity, fat metabolism, bone health, and longevity.
MOTS-c is a mitochondrial-derived peptide (MDP) composed of 16 amino acids, encoded by the 12S rRNA gene of the mitochondrial genome. It plays a critical role in cellular metabolic regulation by modulating glucose metabolism, insulin sensitivity, and cellular stress response through AMPK-dependent pathways.
Overview
MOTS-c was first identified in 2015 by researchers at the USC Leonard Davis School of Gerontology. It represents a class of bioactive peptides derived from short open reading frames within mitochondrial DNA. Unlike most mitochondrial proteins, MOTS-c acts as a retrograde signaling molecule that communicates mitochondrial status to the nucleus and other cellular compartments. Research has shown this peptide to improve exercise capacity, reduce obesity and insulin resistance, and influence disease processes such as osteoporosis and cardiovascular dysfunction.
Mechanism of Action
MOTS-c targets the methionine-folate cycle, increases AICAR (5-aminoimidazole-4-carboxamide ribonucleotide) levels, and activates AMPK, a master regulator of cellular energy homeostasis. AMPK activation drives glucose and fatty acid uptake independently of the insulin signaling pathway. Under metabolic stress, MOTS-c translocates from the cytoplasm to the nucleus, where it regulates genes involved in glucose restriction and antioxidant responses (Kim et al., 2018). This dual-compartment activity positions MOTS-c as a unique mitochondrial-nuclear signaling peptide.
Reconstitution Calculator
MOTS-c
MOTS-c is a mitochondrial-derived peptide (MDP) composed of 16 amino acids, enco

Set up a clean workspace with all supplies ready.
3x / week for weeks
Research
Exercise Mimicry
MOTS-c recapitulates multiple molecular signatures of exercise: AMPK activation, enhanced mitochondrial biogenesis via PGC-1alpha, increased glucose uptake in skeletal muscle, and improved fatty acid oxidation. Kim et al. (2018) showed that exercise increases circulating MOTS-c levels in humans and that MOTS-c administration improves physical capacity in sedentary mice, positioning it as a bona fide exercise-responsive mitokine (Kim et al., 2018).
Aging and Longevity
Circulating MOTS-c levels decline with age in both mice and humans, correlating with metabolic dysfunction and reduced exercise capacity. MOTS-c administration in aged mice restores insulin sensitivity, improves physical performance, and reverses age-dependent metabolic decline. The mitochondrial encoding of MOTS-c means its expression is subject to mitochondrial DNA copy number changes, heteroplasmy, and maternal inheritance patterns, adding genetic complexity to age-related decline.
Obesity and Fat Metabolism
MOTS-c prevents obesity in high-fat diet-fed mice by increasing skeletal muscle fatty acid oxidation and thermogenesis. The S6K1 inhibition mechanism is critical here: S6K1 normally activates lipogenic gene programs via SREBP1c, and MOTS-c-mediated S6K1 suppression shifts the metabolic balance from lipogenesis to lipolysis and beta-oxidation.
Muscle Metabolism
Research in mice indicates that MOTS-c can reverse age-dependent insulin resistance in muscles by improving skeletal muscle response to AMPK activation, which increases the expression of glucose transporters (Lee et al., 2016). This activation is independent of the insulin pathway and offers an alternative means of boosting glucose uptake by muscles when insulin is ineffective or insufficient. The net result is improved muscle function, enhanced muscle growth, and decreased functional insulin resistance.
Insulin Sensitivity
Research measuring MOTS-c levels in insulin-sensitive and insulin-resistant individuals has shown that the peptide is associated with insulin sensitivity only in lean individuals, suggesting MOTS-c may be important in the pathogenesis but not the maintenance of insulin insensitivity (Cataldo et al., 2018). Scientists speculate that changes in MOTS-c levels could act as an early warning sign of potential insulin resistance, and supplementation in this setting could help stave off the development of diabetes.
Osteoporosis
MOTS-c appears to play a role in the synthesis of type I collagen by osteoblasts. Research in osteoblast cell lines shows that MOTS-c regulates the TGF-beta/SMAD pathway responsible for osteoblast health and survival, improving type I collagen synthesis and therefore bone strength and integrity (Che et al., 2019).
Additional research has revealed that MOTS-c promotes differentiation of bone marrow stem cells via the same TGF-beta/SMAD pathway, directly leading to increased osteogenesis (Hu & Chen, 2018). Thus, MOTS-c both protects existing osteoblasts and promotes their development from stem cells.
Longevity
Research has identified a specific variant in the MOTS-c gene (m.1382A>C) that substitutes a glutamate residue for the lysine normally found at position 14. This variant is found exclusively in people with Northeast Asian ancestry and is associated with exceptional longevity in Japanese populations (Fuku et al., 2015). The mechanism by which this substitution affects function remains under investigation, but the structural change likely alters both peptide conformation and receptor interactions.
Metabolic Regulation and Insulin Sensitization
Lee et al. (2015) demonstrated that MOTS-c administration prevents age-dependent and high-fat diet-induced insulin resistance in mice. MOTS-c-treated mice showed improved glucose tolerance, reduced fat accumulation, and increased insulin sensitivity compared to controls. These effects were mediated through AMPK-dependent inhibition of the mTOR/S6K1 axis and enhanced fatty acid oxidation in skeletal muscle (Lee et al., 2015).
Nuclear Gene Regulation
Reynolds et al. (2021) demonstrated that MOTS-c undergoes nuclear translocation under stress conditions (glucose restriction, oxidative stress, exercise). In the nucleus, MOTS-c interacts with chromatin and regulates expression of genes containing antioxidant response elements (AREs), including NRF2-target genes involved in glutathione metabolism, detoxification, and mitochondrial quality control. This finding transformed understanding of MOTS-c from a simple metabolic regulator to a direct transcriptional modulator (Reynolds et al., 2021).
Fat Metabolism
Research in mice has shown that low estrogen levels lead to increased fat mass and adipose tissue dysfunction, increasing the risk of insulin resistance. Supplementing mice with MOTS-c increases brown fat function and reduces adipose tissue accumulation while preventing adipose inflammation that typically precedes insulin resistance (Lu et al., 2019).
MOTS-c's influence on fat metabolism is mediated largely through AMPK pathway activation, which drives fatty acid uptake and oxidation. Research suggests that MOTS-c can leave the mitochondria and translocate to the nucleus where it affects nuclear gene expression, regulating genes involved in glucose restriction and antioxidant responses (Kim et al., 2018). Studies on MOTS-c as a regulator of plasma metabolites have further demonstrated its role in enhancing insulin sensitivity (Kim et al., 2019).
Research has led to a hypothesis that dysregulation of fat metabolism in mitochondria may result in a lack of fat oxidation, leading to higher circulating fat levels and compensatory hyperinsulinemia. MOTS-c supplementation in rats prevents mitochondrial dysfunction and fat accumulation even on a high-fat diet (Crescenzo et al., 2016).
Heart Health
Research measuring MOTS-c levels in humans undergoing coronary angiography has revealed that patients with lower circulating MOTS-c have higher levels of endothelial cell dysfunction. Supplementing rats with MOTS-c sensitizes endothelial cells to signaling molecules like acetylcholine, improving endothelial function and microvascular blood vessel function (Qin et al., 2018).
MOTS-c is not alone among mitochondria-derived peptides in affecting heart health. Research suggests that at least three MDPs play roles in protecting cardiac cells against stress and inflammation, and MDP dysregulation may be an important factor in cardiovascular disease development (Yang et al., 2019).
Safety Profile
MOTS-c is an endogenous peptide naturally produced within human mitochondria, which provides a favorable theoretical safety profile. In animal studies, exogenous MOTS-c administration has not demonstrated significant adverse effects at the doses used in published research. However, no formal human clinical trials have established a comprehensive safety profile. As a metabolically active peptide that influences AMPK signaling, glucose metabolism, and gene expression, potential interactions with diabetes medications and metabolic therapies should be considered. Long-term effects of exogenous supplementation remain uncharacterized.
Animal Study Dosing
Most published MOTS-c research has been conducted in murine models. Lee et al. (2015) used intraperitoneal (IP) injections of MOTS-c at 5 mg/kg/day in C57BL/6 mice fed a high-fat diet for 8 weeks, demonstrating significant reductions in obesity and insulin resistance. Kim et al. (2018) administered 5 mg/kg IP over 12-week periods to characterize nuclear translocation and metabolic gene regulation. In osteoporosis models, Hu & Chen (2018) used 5 mg/kg/day IP in ovariectomized rats for 8 weeks, showing significant bone density improvements.
Exercise and Aging Studies
Kumagai et al. (2022) examined the MOTS-c K14Q variant in exercise physiology, measuring endogenous MOTS-c levels in healthy young adults before and after acute exercise bouts. Plasma MOTS-c levels increased 1.5-2 fold following maximal exercise testing. In aging mouse models, Reynolds et al. (2021) administered 15 mg/kg IP three times weekly for 2 weeks in 22-month-old mice and observed improvements in treadmill endurance, grip strength, and gait performance comparable to younger animals.
Human Observational Data
Cataldo et al. (2018) measured circulating MOTS-c levels in 78 Pima Indian subjects (37 insulin-sensitive, 41 insulin-resistant) using ELISA-based quantification. Mean plasma MOTS-c was significantly higher in lean insulin-sensitive individuals (~0.8 ng/mL) compared to obese insulin-resistant individuals (~0.4 ng/mL). No interventional human clinical trials with exogenous MOTS-c have been published to date.
Pharmacokinetic Profile
MOTS-c — Pharmacokinetic Curve
Subcutaneous injectionQuick Start
- Typical Dose
- Start 5mg daily, increase to 10-15mg based on goals
- Frequency
- Daily for metabolic support, or 3x weekly for anti-aging
- Route
- Subcutaneous injection
- Cycle Length
- 4-12 weeks depending on goals
- Storage
- Refrigerate reconstituted solution at 2-8°C, use within 14 days
Molecular Structure
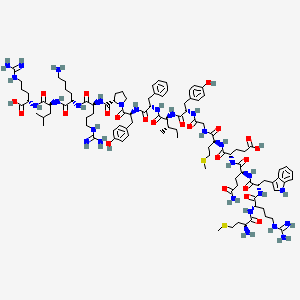
- Formula
- C101H152N28O22S2
- Weight
- 2174.6 Da
- Length
- 16 amino acids
- CAS
- 1627580-64-6
- PubChem CID
- 146675088
- Exact Mass
- 2174.1111 Da
- LogP
- -3.9
- TPSA
- 890 Ų
- H-Bond Donors
- 31
- H-Bond Acceptors
- 29
- Rotatable Bonds
- 73
- Complexity
- 4510
Identifiers (SMILES, InChI)
InChI=1S/C101H152N28O22S2/c1-7-57(4)83(96(148)126-76(50-58-19-9-8-10-20-58)92(144)127-78(52-60-30-34-63(131)35-31-60)97(149)129-46-18-27-79(129)95(147)122-69(25-16-44-112-100(107)108)86(138)118-67(23-13-14-42-102)87(139)124-74(49-56(2)3)91(143)123-73(98(150)151)26-17-45-113-101(109)110)128-94(146)75(51-59-28-32-62(130)33-29-59)116-81(133)55-115-85(137)72(41-48-153-6)121-90(142)71(37-39-82(134)135)119-89(141)70(36-38-80(104)132)120-93(145)77(53-61-54-114-66-22-12-11-21-64(61)66)125-88(140)68(24-15-43-111-99(105)106)117-84(136)65(103)40-47-152-5/h8-12,19-22,28-35,54,56-57,65,67-79,83,114,130-131H,7,13-18,23-27,36-53,55,102-103H2,1-6H3,(H2,104,132)(H,115,137)(H,116,133)(H,117,136)(H,118,138)(H,119,141)(H,120,145)(H,121,142)(H,122,147)(H,123,143)(H,124,139)(H,125,140)(H,126,148)(H,127,144)(H,128,146)(H,134,135)(H,150,151)(H4,105,106,111)(H4,107,108,112)(H4,109,110,113)/t57-,65-,67-,68-,69-,70-,71-,72-,73-,74-,75-,76-,77-,78-,79-,83-/m0/s1
WYTHCOXVWRKRAH-LOKRTKBUSA-NResearch Indications
Metabolic
Improves insulin sensitivity by ~30% in animal studies through AMPK activation.
Restores metabolic function via insulin receptor sensitization.
Promotes fatty acid oxidation despite identical caloric intake.
Anti-Aging
Reverses age-dependent physical decline in animal models.
Enhances mitochondrial function via PGC-1α activation.
6.4% median lifespan extension observed in mice studies.
Exercise Performance
12-15% improvement in running performance from single dose in studies.
Exercise induced 11.9-fold increase in skeletal muscle MOTS-c.
Supports muscle recovery and adaptation to exercise.
Research Protocols
subcutaneous Injection
Mitochondria-derived peptide administered subcutaneously with gradual titration.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Loading phase | 200 mcg | Once daily | Weeks 1-2 |
| Escalation 1 | 400 mcg | Once daily | Weeks 3-4 |
| Escalation 2 | 600 mcg | Once daily | Weeks 5-6 |
| Escalation 3 | 800 mcg | Once daily | Weeks 7-8 |
| Full dose | 1,000 mcg | Once daily | Weeks 9-12(Titrate ~200 mcg every 2 weeks) |
Reconstitution Guide (10mg vial + 3mL BAC water)
- Wipe vial tops with alcohol swab
- Draw 3.0 mL bacteriostatic water into syringe
- Inject slowly down the inside wall of the peptide vial
- Gently swirl to dissolve — never shake
- Resulting concentration: 3.33 mg/mL
- For 200 mcg dose: draw 6 units (0.06 mL)
- For 600 mcg dose: draw 18 units (0.18 mL)
- For 1,000 mcg dose: draw 30 units (0.30 mL)
- Store reconstituted vial refrigerated at 2-8°C
intraperitoneal Injection
- Routes: Intraperitoneal injection (mice); subcutaneous injection anticipated for human translation.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| General Research Protocol | 5 mg | Daily | 7-14 days |
| General Research Protocol | 5 mg | Per protocol | 22 months |
Interactions
Peptide Interactions
MOTS-c's activation of PGC-1alpha and mitochondrial biogenesis programs could synergize with NAD+ precursors (nicotinamide mononucleotide or nicotinamide riboside) that support sirtuin-dependent mitochondrial quality control. Both approaches target age-related mitochondrial decline through comple...
Both are mitochondrial-derived peptides (MDPs) encoded in neighboring regions of the mitochondrial genome (12S vs 16S rRNA). Humanin is primarily cytoprotective (anti-apoptotic, neuroprotective) while MOTS-c is primarily metabolic. Combining two MDPs may recapitulate the full retrograde signaling program of healthy mitochondria.
- Both activate AMPK through distinct upstream mechanisms (MOTS-c via AICAR/folate disruption; spermidine via EP300 inhibition). Additionally, MOTS-c promotes mitochondrial biogenesis (building new mitochondria) while spermidine promotes mitophagy (clearing damaged mitochondria) -- a complementary "build and demolish" approach to mitochondrial quality control.
MOTS-c targets metabolic aging via AMPK/mTOR, while Epithalon targets replicative aging via telomerase. Both undergo nuclear translocation to regulate gene expression, but through different promoter elements (AREs vs immune/circadian gene promoters). Multi-target geroprotection.
What to Expect
What to Expect
AMPK pathway activation; initial glucose tolerance improvements
Enhanced exercise capacity; improved insulin sensitivity
Sustained metabolic benefits; potential body composition changes
Maximum mitochondrial function enhancement; metabolic flexibility
Safety Profile
Common Side Effects
- Generally well-tolerated in animal studies with minimal side effects
- Mild injection site reactions (redness, swelling)
Contraindications
- Pregnancy or breastfeeding
- WADA prohibited substance (banned for athletic competition)
- Limited long-term human safety data
Discontinue If
- Severe allergic reactions or anaphylaxis
- Recurrent hypoglycemia (<50 mg/dL)
- Persistent severe injection site reactions
- Unexplained weight loss >10% of baseline
Quality Indicators
What to look for
- White to off-white powder with uniform appearance indicating proper storage
- Clear, colorless solution without particles after reconstitution
- Sequence verification (MRWQEMGYIFYPRKLR) with CoA >95% purity by RP-HPLC
Caution
- Limited human clinical data - start conservatively
- WADA banned substance - not for competitive athletes
Red flags
- Discolored powder or solution
- Cloudy solution after reconstitution
- No certificate of analysis available
Frequently Asked Questions
References (26)
- [18]Cataldo et al *J Investig Med* J Investig Med (2018)
- [21]
- [1]Obesity and Insulin Sensitivity Study - Lee et al. (2015)
- [2]Physical Performance Across Age Groups - Reynolds et al. (2021)
- [3]Nuclear Translocation Study - Kim et al. (2018)
- [4]Running Performance Study - Hyatt et al. (2022)
- [5]Gestational Diabetes Study - Yin et al. (2022)
- [9]
- [12]
- [19]Che et al *Eur Rev Med Pharmacol Sci* Eur Rev Med Pharmacol Sci (2019)
- [22]Qin et al *Int J Cardiol* Int J Cardiol (2018)
- [23]Yang et al *Biomed Pharmacother* Biomed Pharmacother (2019)
- [16]Kim et al *Physiol Rep* Physiol Rep (2019)
- [17]Crescenzo et al *Eur J Nutr* Eur J Nutr (2016)
- [6]
- [7]
- [8]
- [10]
- [11]
- [27]
- [13]
- [26]
- [14]Lu et al *J Mol Med* J Mol Med (2019)
- [15]Kim et al *Cell Metab* Cell Metab (2018)
- [25]Kumagai et al — The MOTS-c K14Q variant enhances exercise capacity and is associated with longevity iScience (2022)
- [24]
Motilin
Motilin is a 22-amino-acid peptide hormone produced by Mo cells of the duodenal and jejunal mucosa that regulates interdigestive gastrointestinal motility through initiation of the migrating motor complex (MMC). It acts through the motilin receptor (GPR38) and is the physiological basis for erythromycin's prokinetic effects, making it a key target in gastroparesis and motility disorder research.
Nafarelin
Nafarelin is a synthetic GnRH agonist with a D-Nal(2)6 substitution, approximately 200x more potent than native GnRH. Marketed exclusively as an intranasal spray (Synarel, Pfizer), it is FDA-approved for endometriosis and central precocious puberty, and widely used for IVF long-protocol downregulation.