GnRH (Gonadotropin-Releasing Hormone)
GnRH is a hypothalamic decapeptide that serves as the master regulator of vertebrate reproduction, controlling LH and FSH release from the anterior pituitary through pulsatile signaling via the GnRH receptor on gonadotroph cells.
GnRH (Gonadotropin-Releasing Hormone) is a hypothalamic decapeptide that functions as the master regulator of the reproductive endocrine axis. First isolated and characterized by Andrew Schally and Roger Guillemin — work that earned the 1977 Nobel Prize in Physiology or Medicine — GnRH is released in a pulsatile manner from hypothalamic neurons into the hypophyseal portal circulation, where it binds GnRH receptors (GnRHR) on anterior pituitary gonadotroph cells to stimulate synthesis and secretion of luteinizing hormone (LH) and follicle-stimulating hormone (FSH).
Overview
GnRH is the apex signaling molecule of the hypothalamic-pituitary-gonadal (HPG) axis. Synthesized in a distributed network of approximately 1,000-2,000 neurons primarily located in the preoptic area and mediobasal hypothalamus, GnRH neurons integrate upstream signals from kisspeptin/KNDy neurons, metabolic sensors, stress pathways, and circadian inputs to generate coordinated pulsatile release. The frequency and amplitude of GnRH pulses differentially regulate LH and FSH: high-frequency pulses (~1 per 60-90 minutes) favor LH secretion, while lower-frequency pulses favor FSH. Continuous, non-pulsatile GnRH exposure paradoxically suppresses gonadotropin release through receptor downregulation, a phenomenon exploited therapeutically with GnRH agonist drugs.
Mechanism of Action
GnRH binds the GnRH receptor (GnRHR), a Gq/11-coupled GPCR expressed on anterior pituitary gonadotroph cells. Receptor activation triggers phospholipase C-mediated hydrolysis of PIP2, generating IP3 and DAG. IP3 drives intracellular calcium release, while DAG activates protein kinase C, together stimulating LH and FSH synthesis and secretion. Unlike most GPCRs, the GnRHR lacks a cytoplasmic C-terminal tail, which reduces beta-arrestin-mediated desensitization and allows sustained signaling during pulsatile exposure. However, continuous agonist exposure causes receptor internalization and downregulation at the transcriptional level, leading to the paradoxical suppression that defines GnRH agonist pharmacology.
Reconstitution Calculator
GnRH (Gonadotropin-Releasing Hormone)
**GnRH (Gonadotropin-Releasing Hormone)** is a hypothalamic decapeptide that fun

Set up a clean workspace with all supplies ready.
7x / week for weeks
Research
Endometriosis Treatment
LHRH agonists create a hypoestrogenic state that causes regression of endometriotic implants. Henzl et al. (1988) demonstrated that nafarelin nasal spray was effective for endometriosis pain relief. Treatment is typically limited to 6 months due to bone density concerns from prolonged estrogen deprivation, though add-back therapy (low-dose estrogen/progestin) extends the treatment window.
Central Precocious Puberty
LHRH agonists are first-line therapy for central precocious puberty (CPP), suppressing premature activation of the HPG axis to preserve adult height potential and allow age-appropriate psychosocial development. Carel et al. (2009) reviewed long-term outcomes, confirming safety and efficacy of treatment for up to several years with recovery of normal puberty upon discontinuation.
IVF Downregulation Protocols
In assisted reproduction, LHRH agonists are used in the "long protocol" to achieve pituitary downregulation before controlled ovarian stimulation. Starting in the mid-luteal phase of the preceding cycle, daily LHRH agonist administration suppresses endogenous gonadotropin release, preventing premature LH surges during stimulation. The "short/flare" protocol exploits the initial gonadotropin surge to supplement exogenous FSH stimulation Daya (2000).
Prostate Cancer and Androgen Deprivation
The landmark work by Schally et al. (1980) and Labrie et al. (1980) demonstrated that LHRH agonist administration could achieve medical castration equivalent to orchiectomy in men with advanced prostate cancer. This transformed prostate cancer management by providing a reversible, non-surgical alternative. Huggins & Hodges (1941) had established the androgen dependence of prostate cancer decades earlier, but LHRH agonists made androgen deprivation practical and widely accessible.
Pulsatile GnRH and Differential Gonadotropin Regulation
The seminal work by Knobil (1980) demonstrated that pulsatile GnRH delivery is essential for sustained gonadotropin secretion in primates, while continuous infusion leads to suppression. Subsequent studies established that pulse frequency differentially regulates LH versus FSH through distinct transcription factor activation: high-frequency pulses activate Egr-1, which drives LH-beta transcription, while low-frequency pulses activate AP-1, favoring FSH-beta expression Kaiser et al. (1997).
GnRH Receptor Pharmacology
The GnRHR was cloned and characterized in the early 1990s, revealing its unique structural features among GPCRs. Millar et al. (2004) provided a comprehensive review of GnRHR signaling, demonstrating the receptor's coupling to multiple downstream cascades including MAPK/ERK, JNK, and p38 pathways beyond the canonical Gq/11-PLC axis. The absence of the C-terminal tail was shown to confer resistance to rapid desensitization, explaining the requirement for continuous agonist exposure to achieve receptor downregulation.
GnRH in Puberty and Development
GnRH neuron activity is quiescent during childhood and reactivates at puberty onset. Seminara et al. (2003) and de Roux et al. (2003) identified loss-of-function mutations in KISS1R (GPR54) as a cause of hypogonadotropic hypogonadism, establishing the kisspeptin-GnRH pathway as the critical gatekeeper for puberty. GnRH neuron migration from the olfactory placode during embryonic development, when disrupted, causes Kallmann syndrome — the combination of hypogonadotropic hypogonadism and anosmia.
Agonist vs Antagonist Therapeutics
GnRH agonists (leuprolide, goserelin, triptorelin) exploit the paradoxical suppression mechanism: initial flare followed by sustained downregulation of the GnRH receptor, achieving medical castration within 2-4 weeks. GnRH antagonists (cetrorelix, ganirelix, degarelix) block the receptor immediately without flare, providing faster onset of suppression. Conn & Crowley (1994) reviewed the clinical development of both drug classes.
Safety Profile
Endogenous GnRH has an extremely short half-life (2-4 minutes), and exogenous administration (as gonadorelin) produces transient, rapidly reversible effects. Pulsatile GnRH therapy is well-tolerated with minimal side effects beyond injection-site reactions. The safety concerns associated with GnRH pharmacology are primarily related to synthetic agonist and antagonist analogs used in chronic dosing: hot flashes, bone density loss, mood changes, and cardiovascular effects from sustained hypogonadism. Native GnRH itself, when delivered in physiological pulsatile patterns, restores rather than disrupts endocrine function.
Pharmacokinetic Profile
GnRH (Gonadotropin-Releasing Hormone) — Pharmacokinetic Curve
Intravenous injection, Subcutaneous injection, IntranasalQuick Start
- Route
- Intravenous injection, Subcutaneous injection, Intranasal
Molecular Structure
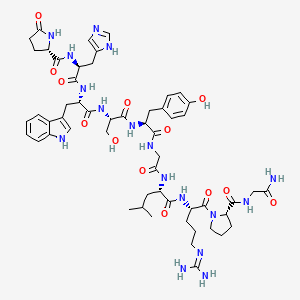
- Formula
- C55H75N17O13
- Weight
- 1182 Da
- CAS
- 33515-09-2
- PubChem CID
- 638793
- Exact Mass
- 1181.5730 Da
- LogP
- -2.4
- TPSA
- 475 Ų
- H-Bond Donors
- 16
- H-Bond Acceptors
- 15
- Rotatable Bonds
- 31
- Complexity
- 2390
Identifiers (SMILES, InChI)
InChI=1S/C55H75N17O13/c1-29(2)19-38(49(80)67-37(9-5-17-60-55(57)58)54(85)72-18-6-10-43(72)53(84)62-25-44(56)75)66-46(77)26-63-47(78)39(20-30-11-13-33(74)14-12-30)68-52(83)42(27-73)71-50(81)40(21-31-23-61-35-8-4-3-7-34(31)35)69-51(82)41(22-32-24-59-28-64-32)70-48(79)36-15-16-45(76)65-36/h3-4,7-8,11-14,23-24,28-29,36-43,61,73-74H,5-6,9-10,15-22,25-27H2,1-2H3,(H2,56,75)(H,59,64)(H,62,84)(H,63,78)(H,65,76)(H,66,77)(H,67,80)(H,68,83)(H,69,82)(H,70,79)(H,71,81)(H4,57,58,60)/t36-,37-,38-,39-,40-,41-,42-,43-/m0/s1
XLXSAKCOAKORKW-AQJXLSMYSA-NResearch Protocols
subcutaneous Injection
Absorption Native GnRH is administered intravenously or subcutaneously.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| General Research Protocol | 5-25 mcg, 100 mcg | Per protocol | 2-4 weeks |
intravenous Injection
Absorption Native GnRH is administered intravenously or subcutaneously.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| General Research Protocol | 5-25 mcg | Per protocol | — |
| General Research Protocol | 100 mcg | Per protocol | — |
intranasal Injection
Intranasal formulations exist but have variable bioavailability (1-3%).
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| General Research Protocol | 5-25 mcg | Per protocol | — |
| General Research Protocol | 100 mcg | Per protocol | — |
| General Research Protocol | 7.5 mg, 22.5 mg, 45 mg, 50 mg, 3.6 mg, 5 mg, 0.5-1.0 mg, 0.25 mg | Daily | 2-4 weeks |
oral
Native LHRH is not orally bioavailable.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| General Research Protocol | 5-25 mcg | Per protocol | — |
| General Research Protocol | 100 mcg | Per protocol | — |
| General Research Protocol | 50 mg | Daily | 2-4 weeks |
| General Research Protocol | 5 mg | Daily | — |
Interactions
Peptide Interactions
Kisspeptin acts as the endogenous activator of GnRH neurons. Dhillo et al. (2005) demonstrated that exogenous kisspeptin administration produces GnRH-dependent LH release. Combined assessment using both kisspeptin and GnRH challenge tests allows differentiation of hypothalamic vs. pituitary cause...
What to Expect
What to Expect
Rapid onset expected; half-life of 2-4 minutes (IV) indicates fast-acting pharmacokinetics
The GnRH stimulation test involves a single IV bolus of gonadorelin (100 mcg) with LH and FSH measurements at 0, 30, and 60 minutes.
Within 1-2 weeks of continuous agonist exposure, LH and FSH fall to castrate levels — a phenomenon called downregulation that is exploited...
GnRH agonists (leuprolide, goserelin, triptorelin) exploit the paradoxical suppression mechanism: initial flare followed by sustained downregulation...
Continued use as directed
Quality Indicators
What to look for
- Well-established safety profile
- Extensive peer-reviewed research base
- Oral administration available
Caution
- Short half-life may require frequent dosing
- Injection site reactions reported
Frequently Asked Questions
References (10)
- [10]
- [12]Millar & Newton — Current and future applications of GnRH, kisspeptin, and neurokinin B Nat. Rev. Endocrinol. (2023)
- [1]Schally et al *Science*, 173(4001), 1036-1038 Science (1971)
- [2]Knobil *Recent Prog Recent Prog. Horm. Res. (1980)
- [4]Kaiser et al *Proc Proc. Natl. Acad. Sci. (1997)
- [11]
- [3]Conn & Crowley *N N. Engl. J. Med. (1994)
- [8]Schally *Peptides*, 20(10), 1247-1262 Peptides (1999)
- [5]
- [9]Martin et al *J J. Clin. Endocrinol. Metab. (1990)
Glucagon
Glucagon is a 29-amino acid counter-regulatory peptide hormone produced by pancreatic alpha cells that raises blood glucose through hepatic glycogenolysis and gluconeogenesis. It is used therapeutically for severe hypoglycemia, GI imaging, and beta-blocker/calcium channel blocker overdose, with emerging research into dual GLP-1/glucagon receptor agonists for obesity and metabolic disease.
Gonadorelin
Gonadorelin (GnRH) is a decapeptide gonadotropin-releasing hormone that stimulates LH and FSH release, with clinical applications in fertility assessment and research into breast cancer, prostate cancer, and Alzheimer's disease.