Semaglutide
Semaglutide is a GLP-1 receptor agonist that mimics the incretin hormone GLP-1 to regulate blood sugar, reduce appetite, and promote weight loss. It is widely researched for type 2 diabetes, obesity, cardiovascular risk reduction, and emerging neurological applications.
Semaglutide is a synthetic glucagon-like peptide-1 (GLP-1) receptor agonist and analogue of human GLP-1, a hormone naturally produced in the intestinal L-cells that regulates blood sugar levels after meals. It is one of the most extensively studied peptides for type 2 diabetes mellitus and obesity, with emerging research into cardiovascular protection, neurodegeneration, and addiction.
Overview
Semaglutide works by mimicking the action of GLP-1 to stimulate insulin release, reduce glucagon secretion, slow gastric emptying, and promote satiety. Its extended half-life of approximately one week (achieved through albumin binding via a fatty acid side chain) allows once-weekly dosing for injectable formulations. Semaglutide is marketed under brand names including Ozempic (diabetes), Wegovy (obesity), and Rybelsus (oral formulation for diabetes).
GLP-1 (Glucagon-Like Peptide-1) is an incretin hormone released by intestinal L-cells in response to food intake. It stimulates glucose-dependent insulin secretion, suppresses glucagon release, slows gastric emptying, and signals satiety to the brain. GLP-1 receptor agonists like semaglutide leverage these mechanisms with enhanced pharmacokinetic stability compared to native GLP-1, which has a half-life of only 2-3 minutes.
Mechanism of Action
Semaglutide activates GLP-1 receptors through several coordinated physiological pathways:
Insulin Secretion: Binding to GLP-1 receptors on pancreatic beta cells stimulates glucose-dependent insulin release, reducing blood sugar levels only when they are elevated.
Glucagon Suppression: Semaglutide suppresses glucagon secretion from pancreatic alpha cells, reducing hepatic glucose output.
Gastric Emptying: The peptide slows the rate at which the stomach empties into the small intestine, stabilizing post-meal glucose excursions and contributing to satiety.
Central Appetite Regulation: Semaglutide acts on GLP-1 receptors in the hypothalamus and brainstem to reduce appetite and food intake. There is growing evidence it also reduces cravings and modulates reward-related feeding behavior.
Beta Cell Protection: GLP-1 receptor activation promotes beta cell proliferation and inhibits apoptosis, potentially preserving pancreatic function over time in type 2 diabetes.
Reconstitution Calculator
Reconstitution Calculator
Calculate your peptide dosing

Set up a clean workspace with all supplies ready.
7x / week for weeks
Research
PIONEER 10 — Japanese Population
PIONEER 10 evaluated oral semaglutide in Japanese patients with type 2 diabetes, comparing it with injectable dulaglutide 0.75 mg. Results were consistent with the global program, with oral semaglutide 14 mg achieving comparable or superior glycemic control and weight loss. Yabe, D. et al. (2020) — Diabetes Obes. Metab.
PIONEER 4 — vs. Liraglutide
PIONEER 4 compared oral semaglutide 14 mg with injectable liraglutide 1.8 mg and placebo. At 26 weeks, oral semaglutide was non-inferior to liraglutide for HbA1c reduction and superior for weight loss (4.4 kg vs. 3.1 kg vs. 0.5 kg for placebo). This trial was critical for demonstrating that an oral GLP-1 agonist could match or exceed the efficacy of a standard injectable GLP-1 agonist. Pratley, R. E. et al. (2019) — Lancet
PIONEER 7 — Flexible Dose Adjustment
PIONEER 7 compared oral semaglutide with flexible dose adjustment (3, 7, or 14 mg based on glycemic response and tolerability) versus sitagliptin 100 mg. The flexible dosing approach demonstrated superior HbA1c reduction and weight loss compared to sitagliptin, providing evidence for individualized dosing strategies. Pieber, T. R. et al. (2019) — Lancet Diabetes Endocrinol.
PIONEER 2 — vs. Empagliflozin
PIONEER 2 compared oral semaglutide 14 mg with the SGLT2 inhibitor empagliflozin 25 mg in patients with type 2 diabetes inadequately controlled on metformin. At 26 weeks, oral semaglutide achieved superior HbA1c reduction (1.3% vs. 0.9%) and comparable weight loss. At 52 weeks, the HbA1c advantage was maintained. This trial positioned oral semaglutide as a competitive alternative to SGLT2 inhibitors in the second-line treatment setting. Rodbard, H. W. et al. (2019) — JAMA
PIONEER 3 — vs. Sitagliptin
PIONEER 3 compared oral semaglutide (3, 7, and 14 mg) with the DPP-4 inhibitor sitagliptin 100 mg in patients with type 2 diabetes on metformin with or without a sulfonylurea. At 26 weeks, oral semaglutide 7 and 14 mg demonstrated superior HbA1c reduction compared to sitagliptin (1.0% and 1.3% vs. 0.8%). Weight loss was significantly greater with oral semaglutide. The trial established superiority over DPP-4 inhibitors, the most commonly prescribed oral diabetes class. Rosenstock, J. et al. (2019) — JAMA
PIONEER 5 — Renal Impairment
PIONEER 5 evaluated oral semaglutide 14 mg in patients with type 2 diabetes and moderate renal impairment (eGFR 30-59 mL/min/1.73m2). The trial demonstrated efficacy and safety consistent with the broader population, with HbA1c reduction of 1.0% and weight loss of 3.4 kg at 26 weeks. No significant renal safety concerns were identified. Mosenzon, O. et al. (2019) — Lancet Diabetes Endocrinol.
PIONEER 6 — Cardiovascular Safety
PIONEER 6 was a cardiovascular outcomes trial (CVOT) designed to demonstrate cardiovascular safety (non-inferiority to placebo for MACE). The trial enrolled 3,183 patients with type 2 diabetes at high cardiovascular risk. After a median follow-up of 15.9 months, oral semaglutide met its primary non-inferiority endpoint (HR 0.79; 95% CI 0.57-1.11; p<0.001 for non-inferiority). A numerical but non-significant reduction in MACE was observed. Cardiovascular death was significantly lower with oral semaglutide (HR 0.49; 95% CI 0.27-0.92), though the trial was not powered for this endpoint. Husain, M. et al. (2019) — N. Engl. J. Med.
Weight Loss
Research indicates that semaglutide produces significant weight loss through reduced appetite, slowed gastric emptying, decreased insulin resistance, and shifts away from fat storage. In the STEP 1 trial, participants receiving semaglutide 2.4 mg weekly achieved a mean weight reduction of 14.9% over 68 weeks compared to 2.4% with placebo. Wilding et al. (2021) — N. Engl. J. Med.
Cardiovascular Outcomes
The SUSTAIN-6 trial demonstrated that semaglutide significantly reduced the risk of major adverse cardiovascular events (MACE) in patients with type 2 diabetes at high cardiovascular risk, with a 26% reduction in the composite endpoint of cardiovascular death, nonfatal myocardial infarction, and nonfatal stroke. Marso et al. (2016) — N. Engl. J. Med.
PIONEER 8 — Add-On to Insulin
PIONEER 8 evaluated oral semaglutide added to insulin therapy (with or without metformin) in type 2 diabetes. Oral semaglutide 14 mg reduced HbA1c by 1.3% and body weight by 3.3 kg versus placebo at 26 weeks, while enabling insulin dose reduction. Zinman, B. et al. (2019) — Diabetes Care
Fatigue and Sleep
Semaglutide causes fatigue in a subset of users (0.4-11% depending on formulation), primarily through calorie deficit and potentially through central GLP-1 signaling mechanisms. GLP-1 release is biphasic, and the second phase (30-60 minutes post-meal) may contribute to postprandial sleepiness. Researchers are also investigating GLP-1 receptor agonists for sleep apnea, with emerging evidence that they may directly impact non-rapid eye movement sleep duration beyond weight-loss-mediated benefits.
Comparison with Sermorelin
Both semaglutide and sermorelin alter body composition and promote fat burning, but through fundamentally different mechanisms. Sermorelin stimulates growth hormone release from the pituitary via GHRH receptor activation, while semaglutide acts through GLP-1 receptor-mediated appetite suppression and metabolic regulation. The choice between them depends on clinical context and whether the primary goal is GH axis restoration or appetite/glucose control.
PIONEER 1 — Monotherapy
The PIONEER 1 trial was a 26-week, randomized, double-blind, placebo-controlled study evaluating oral semaglutide 3, 7, and 14 mg as monotherapy in treatment-naive patients with type 2 diabetes. Oral semaglutide 14 mg demonstrated a statistically significant reduction in HbA1c of 1.5 percentage points from baseline (vs. 0.0% for placebo) and a mean body weight reduction of 3.7 kg. The trial established oral semaglutide's efficacy as first-line monotherapy. Aroda, V. R. et al. (2019) — Lancet
Higher Oral Doses (25 mg, 50 mg)
Novo Nordisk has investigated higher oral semaglutide doses (25 mg and 50 mg daily) to achieve efficacy more closely matching injectable semaglutide 2.4 mg for weight management. The OASIS 1 trial evaluated oral semaglutide 50 mg for weight management in adults with overweight or obesity without diabetes. At 68 weeks, oral semaglutide 50 mg achieved a mean weight loss of approximately 15.1%, approaching the 14.9% achieved with injectable semaglutide 2.4 mg in STEP 1. This represents a significant advancement toward an all-oral alternative to injectable GLP-1 agonist therapy for obesity. Knop, F. K. et al. (2023) — Lancet
Safety Profile
Common adverse effects include nausea (15-20%), diarrhea, vomiting, and constipation, which are typically mild to moderate and diminish over time with dose escalation. Injection site reactions occur infrequently. Semaglutide carries warnings for thyroid C-cell tumors (based on rodent studies; relevance to humans uncertain), pancreatitis, gallbladder disease, and hypoglycemia when used with insulin or sulfonylureas. It is contraindicated in patients with a personal or family history of medullary thyroid carcinoma or Multiple Endocrine Neoplasia syndrome type 2 (MEN 2). Fatigue, reported in 0.4-11% of users depending on formulation, is typically self-limiting. Rare cases of acute kidney injury have been reported, usually in the context of dehydration from gastrointestinal side effects.
Pharmacokinetic Profile
Semaglutide — Pharmacokinetic Curve
Subcutaneous injection (pre-filled pen)Ongoing & Future Research
- Higher-dose oral formulations: Oral semaglutide 25 mg and 50 mg are under regulatory review for enhanced glycemic control and weight management. The 50 mg dose demonstrated weight loss (~15%) approaching injectable semaglutide 2.4 mg.
- Obesity indication for oral semaglutide: Based on OASIS 1 results, regulatory submissions for an obesity indication (likely 50 mg dose) are anticipated.
- Next-generation oral delivery: Research into improved absorption enhancers and formulation technologies to increase oral bioavailability beyond 1%, which could enable lower doses and improved dose-response predictability.
- Oral peptide combinations: Investigation of oral co-formulations combining semaglutide with other peptide therapeutics (e.g., amylin analogues).
- Cardiovascular outcome superiority: A larger, adequately powered CVOT for oral semaglutide could establish cardiovascular superiority, as PIONEER 6 showed numerical but non-significant MACE reduction.
- NASH/MASH: Evaluation of higher-dose oral semaglutide in metabolic-associated steatohepatitis.
- Patient preference and adherence: Real-world studies comparing adherence, persistence, and quality of life between oral and injectable semaglutide formulations.
Quick Start
- Typical Dose
- 0.25 mg starting, titrate to 1-2.4 mg weekly
- Frequency
- Once weekly (injectable) or once daily (oral)
- Route
- Subcutaneous injection (pre-filled pen)
- Timing
- Same day each week, any time of day. Oral: first thing in morning on empty stomach.
- Cycle Length
- Ongoing therapy (not cycled)
- Break Between Cycles
- N/A — continuous use; weight regain occurs upon discontinuation
- Storage
- Refrigerate 2-8°C before first use. In-use pen at room temp up to 6 weeks.
Molecular Structure
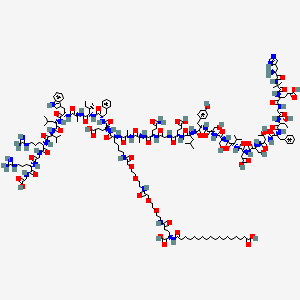
- Formula
- C187H291N45O59
- Weight
- 4113.58 Da
- Length
- 31 amino acids
- CAS
- 910463-68-2
- PubChem CID
- 56843331
- Exact Mass
- 4112.1187 Da
- LogP
- -5.8
- TPSA
- 1650 Ų
- H-Bond Donors
- 57
- H-Bond Acceptors
- 63
- Rotatable Bonds
- 151
- Complexity
- 9590
Identifiers (SMILES, InChI)
InChI=1S/C187H291N45O59/c1-18-105(10)154(180(282)208-108(13)159(261)216-133(86-114-89-200-119-50-40-39-49-117(114)119)170(272)218-129(82-102(4)5)171(273)228-152(103(6)7)178(280)215-121(53-44-72-199-186(192)193)162(264)201-91-141(242)209-120(52-43-71-198-185(190)191)161(263)204-94-151(257)258)230-172(274)131(83-111-45-33-31-34-46-111)219-167(269)126(64-69-149(253)254)214-166(268)122(51-41-42-70-195-144(245)98-290-79-78-289-76-74-197-145(246)99-291-80-77-288-75-73-196-139(240)66-61-127(183(285)286)211-140(241)54-37-29-27-25-23-21-19-20-22-24-26-28-30-38-55-146(247)248)212-158(260)107(12)206-157(259)106(11)207-165(267)125(60-65-138(189)239)210-142(243)92-202-163(265)123(62-67-147(249)250)213-168(270)128(81-101(2)3)217-169(271)130(85-113-56-58-116(238)59-57-113)220-175(277)135(95-233)223-177(279)137(97-235)224-179(281)153(104(8)9)229-174(276)134(88-150(255)256)221-176(278)136(96-234)225-182(284)156(110(15)237)231-173(275)132(84-112-47-35-32-36-48-112)222-181(283)155(109(14)236)227-143(244)93-203-164(266)124(63-68-148(251)252)226-184(287)187(16,17)232-160(262)118(188)87-115-90-194-100-205-115/h31-36,39-40,45-50,56-59,89-90,100-110,118,120-137,152-156,200,233-238H,18-30,37-38,41-44,51-55,60-88,91-99,188H2,1-17H3,(H2,189,239)(H,194,205)(H,195,245)(H,196,240)(H,197,246)(H,201,264)(H,202,265)(H,203,266)(H,204,263)(H,206,259)(H,207,267)(H,208,282)(H,209,242)(H,210,243)(H,211,241)(H,212,260)(H,213,270)(H,214,268)(H,215,280)(H,216,261)(H,217,271)(H,218,272)(H,219,269)(H,220,277)(H,221,278)(H,222,283)(H,223,279)(H,224,281)(H,225,284)(H,226,287)(H,227,244)(H,228,273)(H,229,276)(H,230,274)(H,231,275)(H,232,262)(H,247,248)(H,249,250)(H,251,252)(H,253,254)(H,255,256)(H,257,258)(H,285,286)(H4,190,191,198)(H4,192,193,199)/t105-,106-,107-,108-,109+,110+,118-,120-,121-,122-,123-,124-,125-,126-,127+,128-,129-,130-,131-,132-,133-,134-,135-,136-,137-,152-,153-,154-,155-,156-/m0/s1
DLSWIYLPEUIQAV-CCUURXOWSA-NResearch Indications
Diabetes
FDA-approved for type 2 diabetes. SUSTAIN trials demonstrated HbA1c reductions of 1.5-2.0% with injectable formulations.
SUSTAIN-6 showed 26% reduction in composite of cardiovascular death, nonfatal MI, and nonfatal stroke in high-risk diabetic patients.
Promotes beta cell proliferation and inhibits apoptosis, potentially preserving pancreatic function long-term.
Neurological
Multiple trials investigating semaglutide for cognitive outcomes in early Alzheimer's, based on GLP-1R expression in the brain and preclinical neuroprotection data.
Preclinical evidence of reduced alcohol intake via GLP-1R signaling in brain reward circuits. Phase 2 human trials underway.
Other
STEP trials evaluating weight-loss-mediated and potentially direct GLP-1R-mediated improvements in sleep apnea.
Emerging data suggests renal protective effects beyond glucose and weight management. Dedicated renal outcome trials in progress.
Weight Loss
FDA-approved for chronic weight management in adults with BMI ≥30 or ≥27 with comorbidities. STEP 1 demonstrated 14.9% mean weight loss at 68 weeks.
Reduces hunger and food cravings through central nervous system GLP-1 receptor activation in hypothalamus and brainstem.
STEP 5 demonstrated sustained 15.2% weight loss at 104 weeks with continued treatment.
STEP TEENS showed 16.1% BMI reduction in adolescents aged 12-17 over 68 weeks.
Central GLP-1 receptor activation in hypothalamus reduces hunger and food cravings. PIONEER trials showed consistent weight loss of 3-5 kg at approved diabetes doses.
Oral semaglutide reduces visceral adiposity as demonstrated by imaging substudies, contributing to metabolic improvements beyond scale weight.
Cardiovascular
SELECT trial demonstrated 20% reduction in major adverse cardiovascular events in overweight/obese patients without diabetes over 5 years.
STEP-HFpEF showed improved symptoms, physical limitations, and weight in HFpEF patients with obesity.
SOUL trial demonstrated significant reduction in major adverse cardiovascular events (cardiovascular death, nonfatal MI, nonfatal stroke) in T2DM patients with established cardiovascular disease.
PIONEER trials showed systolic blood pressure reductions of 2-5 mmHg, likely mediated by weight loss and direct vascular GLP-1R effects.
Modest improvements in triglycerides and LDL cholesterol observed across PIONEER trials, contributing to overall cardiovascular risk reduction.
Metabolic
Addresses multiple components simultaneously: weight, glucose, blood pressure, and lipid profiles.
Newsome et al. (2021) showed semaglutide improved NASH resolution without worsening fibrosis. Phase 3 trials ongoing.
Decreases systemic inflammation markers through mechanisms partly independent of weight loss.
FDA-approved (Rybelsus) for T2DM. PIONEER trials demonstrated HbA1c reductions of 1.0-1.4% across doses (7mg and 14mg), comparable to injectable semaglutide in PIONEER 9.
PIONEER 1 showed significant fasting plasma glucose reductions of 1.2-1.6 mmol/L versus placebo at 26 weeks with oral semaglutide 14mg.
GLP-1 receptor agonism enhances glucose-dependent insulin secretion and suppresses inappropriate glucagon release, supporting residual beta cell function in T2DM.
Research Protocols
subcutaneous Injection
GLP-1 receptor agonist administered once weekly with slow dose escalation to minimize GI side effects.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Initiation | 250 mcg | Once weekly | Weeks 1-4 |
| Escalation 1 | 500 mcg | Once weekly | Weeks 5-8 |
| Escalation 2 | 1,000 mcg | Once weekly | Weeks 9-12 |
| Escalation 3 | 1,700 mcg | Once weekly | Weeks 13-16 |
| Maintenance | 2,400 mcg | Once weekly | Week 17+(Continue at maintenance indefinitely) |
Reconstitution Guide (5mg vial + 2mL BAC water)
- Wipe vial tops with alcohol swab
- Draw 2.0 mL bacteriostatic water into syringe
- Inject slowly down the inside wall of the peptide vial
- Gently swirl to dissolve — never shake
- Resulting concentration: 2.5 mg/mL
- For 250 mcg dose: draw 10 units (0.10 mL)
- For 500 mcg dose: draw 20 units (0.20 mL)
- For 1,000 mcg dose: draw 40 units (0.40 mL)
- For 2,400 mcg dose: draw 96 units (0.96 mL)
- Store reconstituted vial refrigerated at 2-8°C
oral
Oral semaglutide (marketed as Rybelsus) is the first oral formulation of a glucagon-like peptide-1 (GLP-1) receptor agonist approved for clinical use. Overview The development of oral semaglutide addressed one of the most significant barriers to GLP-1 agonist adoption: the requirement for subcutane
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Dosing (Rybelsus — approved) | 3 mg, 7 mg, 14 mg | Daily | 30 days(Route: Oral) |
| Dosing (higher doses — investigational) | 25 mg, 50 mg | Daily | — |
| Key trials | 50 mg, 25 mg | Per protocol | — |
| Treatment-naive T2D | 14 mg | Per protocol | — |
| Overweight/obesity without diabetes | 1 — 50 mg | Per protocol | 68 weeks(Route: Oral) |
| Higher-dose oral formulations | 25 mg, 50 mg, 2.4 mg | Per protocol | —(Route: Oral) |
| Obesity indication for oral semaglutide | 50 mg | Per protocol | —(Route: Oral) |
| Administration requirements | 14 mg, 100 mg | Per protocol | —(Route: Oral) |
| Diabetes initiation | 3 mg | Once daily | 30 days then increase(Take on empty stomach with ≤4 oz water, 30 min before food/other meds) |
| Standard diabetes dose | 7 mg | Once daily | Ongoing(Increase from 3 mg after 30 days) |
| Maximum diabetes dose | 14 mg | Once daily | Ongoing(May increase from 7 mg for additional glycemic control) |
Interactions
Peptide Interactions
Both semaglutide and sermorelin alter body composition and promote fat burning, but through fundamentally different mechanisms. Sermorelin stimulates growth hormone release from the pituitary via GHRH receptor activation, while semaglutide acts through GLP-1 receptor-mediated appetite suppression...
What to Expect
What to Expect
Mild appetite reduction during initial low dose (0.25 mg). Possible nausea as body adjusts. Blood sugar improvements may begin within 1-2 weeks in diabetic patients.
Noticeable weight loss (5-10% typical). Improved satiety and reduced food cravings. Dose escalation continues through this period.
Significant weight loss (10-15% common). Stable glucose levels in diabetic patients. Reach maintenance dose (2.4 mg for weight loss).
Continued weight loss approaching peak effect (14.9% mean at 68 weeks in STEP 1). Cardiovascular and metabolic biomarker improvements.
Sustained weight maintenance with continued treatment (15.2% at 104 weeks in STEP 5). Weight regain of ~2/3 of lost weight within one year if discontinued.
Safety Profile
Common Side Effects
- Nausea (15-20%, typically improves with dose escalation)
- Diarrhea
- Vomiting
- Constipation
- Abdominal pain
- Fatigue (0.4-11% depending on formulation)
- Injection site reactions (infrequent)
Contraindications
- Personal or family history of medullary thyroid carcinoma (MTC)
- Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
- Pregnancy or breastfeeding
- History of pancreatitis (use with caution)
- Concurrent use with other GLP-1 receptor agonists
Discontinue If
- Severe persistent abdominal pain radiating to back (possible pancreatitis)
- Persistent vomiting preventing adequate hydration
- Neck mass, hoarseness, or difficulty swallowing (thyroid tumor signs)
- Severe allergic reaction (rash, swelling, difficulty breathing)
- Vision changes (possible diabetic retinopathy progression)
- Signs of acute gallbladder disease
- Worsening renal function or decreased urination
Drug Interactions
- Insulin: increased hypoglycemia risk — insulin dose reduction typically needed
- Sulfonylureas: increased hypoglycemia risk — dose reduction may be required
- Oral medications: delayed gastric emptying may alter absorption timing of co-administered oral drugs
- Warfarin: monitor INR more frequently when initiating semaglutide due to potential absorption changes
- Oral contraceptives: delayed gastric emptying may reduce absorption — consider non-oral alternatives
Uncommon
- Headache
- Dizziness
- Bloating and flatulence
- Gastroesophageal reflux
- Brain fog or cognitive sluggishness
- Hair thinning (anecdotal, likely related to rapid weight loss)
Serious
- Pancreatitis (monitor for severe persistent abdominal pain)
- Thyroid C-cell tumors (observed in rodent studies; human relevance uncertain)
- Diabetic retinopathy progression
- Acute kidney injury (usually in context of dehydration from GI side effects)
- Gallbladder disease (cholelithiasis, cholecystitis)
- Severe hypoglycemia (when combined with insulin or sulfonylureas)
- Hypomagnesemia and hypocalcemia (rare, especially with polypharmacy)
Quality Indicators
What to look for
- FDA-approved branded products (Ozempic, Wegovy, Rybelsus) from licensed pharmacies
- Clear, colorless to slightly yellow solution (injectable)
- Proper cold-chain storage verified and product not expired
- Pre-filled pen with intact seal and visible lot number
- Third-party purity certificate if compounded (HPLC ≥98%)
Caution
- Compounded semaglutide — FDA has warned about untested compounded versions
- Product stored outside recommended temperature range
- Approaching expiration date
Red flags
- Cloudy or discolored solution
- Visible particles or precipitation
- Non-pharmacy or unverified online sources
- No lot number, expiration date, or COA available
- Broken cold chain (left unrefrigerated before first use)
Frequently Asked Questions
References (11)
- [8]Davies M, Pieber TR, Hartoft-Nielsen ML, et al. Effect of Oral Semaglutide Compared With Placebo and Subcutaneous Semaglutide on Glycemic Control in Patients With Type 2 Diabetes (PIONEER 1) JAMA (2017)
→ Oral semaglutide demonstrated clinically meaningful HbA1c reductions in the PIONEER program (n=9,543)
- [10]Real-World Safety Analysis Long-term Real-World Safety of Semaglutide in 175,000 Patients Diabetes Care (2025)
→ Analysis of 175,000 patients confirms long-term safety with no new safety signals identified
- [11]STEP UP Investigators Higher-Dose Semaglutide (7.2 mg) for Weight Loss (STEP UP) Nature Medicine (2024)
→ 7.2 mg weekly dose achieved 20.7% weight loss at 72 weeks
- [12]CagriSema Phase 2 Investigators Cagrilintide Plus Semaglutide for Weight Management (CagriSema Phase 2) The Lancet (2024)
→ Combination of cagrilintide + semaglutide achieved 25% weight loss in Phase 2 trial
- [1]Wilding JPH, Batterham RL, Calanna S, et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity (STEP 1) New England Journal of Medicine (2021)
→ 14.9% mean weight loss with semaglutide 2.4 mg weekly vs 2.4% with placebo at 68 weeks in 1,961 participants
- [2]Marso SP, Bain SC, Consoli A, et al. Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes (SUSTAIN-6) New England Journal of Medicine (2016)
→ 26% reduction in MACE composite in T2D patients at high cardiovascular risk
- [3]Lincoff AM, Brown-Frandsen K, Colhoun HM, et al. Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes (SELECT) New England Journal of Medicine (2023)
→ 20% reduction in MACE in overweight/obese patients without diabetes over 5 years (n=17,604)
- [4]Rubino DM, Greenway FL, Khalid U, et al. Effect of Continued Weekly Subcutaneous Semaglutide vs Placebo on Weight Loss Maintenance (STEP 5) JAMA (2024)
→ Sustained 15.2% weight loss at 104 weeks with continued semaglutide 2.4 mg weekly
- [5]Kosiborod MN, Abildstrom SZ, Borlaug BA, et al. Semaglutide in Patients with Heart Failure with Preserved Ejection Fraction and Obesity (STEP-HFpEF) New England Journal of Medicine (2023)
→ Improved symptoms, physical limitations, and weight in HFpEF patients with obesity
- [6]Newsome PN, Buchholtz K, Cusi K, et al. A Placebo-Controlled Trial of Subcutaneous Semaglutide in Nonalcoholic Steatohepatitis New England Journal of Medicine (2021)
→ Semaglutide improved NASH resolution without worsening fibrosis
- [7]Weghuber D, Barrett T, Barrientos-Perez M, et al. Once-Weekly Semaglutide in Adolescents with Obesity (STEP TEENS) New England Journal of Medicine (2022)
→ 16.1% BMI reduction in adolescents aged 12-17 over 68 weeks
Selank
Selank is a synthetic heptapeptide analogue of the immunomodulatory peptide tuftsin, developed at the Institute of Molecular Genetics of the Russian Academy of Sciences as an anxiolytic nootropic with GABAergic and monoaminergic activity.
Semax
Semax is a synthetic heptapeptide derived from adrenocorticotropic hormone (ACTH) fragments 4-10 that increases BDNF and NGF levels in the brain. It is studied for neuroprotection, cognitive enhancement, stroke recovery, and antidepressant effects.