TB-500
TB-500 is a synthetic fragment of thymosin beta-4, a 43-amino acid protein involved in actin regulation, tissue repair, wound healing, and anti-inflammatory signaling across multiple organ systems.
TB-500 is a synthetic peptide derived from the active region of thymosin beta-4 (TB4), a naturally occurring 43-amino acid protein found in virtually all human and animal cells. TB-500 corresponds to the actin-binding domain of TB4 and retains its key biological activities, including promotion of cell migration, angiogenesis, and tissue repair.
Overview
Thymosin beta-4 was originally isolated from the thymus gland in 1981 by Allan Goldstein and colleagues and is one of the most abundant intracellular proteins in eukaryotic cells. Its primary known function is the sequestration of monomeric actin (G-actin), regulating the polymerization of actin filaments that are essential for cell motility, division, and structural integrity. TB-500, the synthetic version used in research, encompasses the 17-amino acid active region of TB4 responsible for actin binding and many of its downstream biological effects.
TB-500 has gained significant attention in both research and veterinary contexts for its ability to accelerate wound healing, reduce inflammation, and promote regeneration of damaged tissues including muscle, tendon, ligament, and cardiac tissue. In veterinary medicine, particularly in equine practice, TB4 has been widely used to treat soft tissue injuries and promote recovery from musculoskeletal damage.
Mechanism of Action
TB-500 exerts its effects through several interconnected pathways:
Actin Sequestration and Regulation: TB-500 binds to G-actin through its central LKKTETQ motif, regulating the balance between monomeric G-actin and filamentous F-actin. This process is critical for cell migration, a fundamental requirement for wound healing and tissue repair. By modulating actin polymerization dynamics, TB-500 enhances the motility of endothelial cells, keratinocytes, and other repair-associated cell types. Safer et al. (1997) determined the crystal structure of the TB4-actin complex, revealing that the peptide wraps around the actin monomer to prevent polymerization until needed.
Angiogenesis: TB-500 stimulates the formation of new blood vessels by promoting endothelial cell migration and tubule formation. This neovascularization improves blood supply to damaged tissues, accelerating nutrient delivery and waste removal necessary for healing. Grant et al. (1999) demonstrated that TB4 promotes angiogenesis both in vitro and in the chick chorioallantoic membrane assay.
Anti-inflammatory Signaling: TB-500 downregulates pro-inflammatory cytokines including TNF-alpha, IL-1beta, and MIP-2, while promoting anti-inflammatory mediators. This dual action helps resolve acute inflammation and prevents chronic inflammatory damage. These effects appear to be mediated through suppression of NF-kappaB signaling and modulation of inflammasome activity. Sosne et al. (2007) showed that TB4 modulates inflammatory mediators in corneal epithelial cells.
Matrix Metalloproteinase Regulation: TB-500 modulates the activity of matrix metalloproteinases (MMPs), enzymes involved in extracellular matrix remodeling. This regulation is essential for orderly tissue repair and prevention of excessive scarring.
Cardiac Progenitor Cell Activation: In cardiac tissue, TB4 activates epicardium-derived progenitor cells (EPDCs), promoting their migration to sites of ischemic injury where they differentiate into cardiomyocytes and vascular smooth muscle cells. Smart et al. (2007) demonstrated that TB4 reactivates the embryonic epicardial developmental program in adult hearts.
Reconstitution Calculator
TB-500
**TB-500** is a synthetic peptide derived from the active region of thymosin bet
Exceeds syringe capacity
Dose requires 2.000mL but syringe holds 1mL. Increase BAC water, use a larger syringe, or split injections.

Set up a clean workspace with all supplies ready.
2x / week for weeks
Research
Comparison with Systemic TB-500 Administration
Systemic TB-500 (subcutaneous or intraperitoneal injection) distributes broadly across peripheral tissues and crosses the BBB to a limited extent. While this route has demonstrated neuroprotective effects in animal studies, brain concentrations are a fraction of peripheral levels. Intranasal delivery theoretically achieves higher brain-to-plasma ratios, concentrating the peptide where it is needed for CNS applications. However, systemic TB-500 remains advantageous for peripheral tissue repair (wound healing, cardiac protection, musculoskeletal injury) where peripheral distribution is desirable.
Traumatic Brain Injury
Xiong et al. demonstrated that systemic TB4 administration following controlled cortical impact in rats reduced neuronal cell death, promoted oligodendrocyte differentiation, and enhanced axonal remodeling. TB4-treated animals showed improved spatial learning and sensorimotor function compared to saline controls. Intranasal delivery of TB4 in TBI models builds on these findings by targeting higher brain concentrations. The peptide's multi-modal mechanism — combining neuroprotection, anti-inflammation, and neurogenesis — makes it particularly suited for the complex pathophysiology of TBI, which involves primary mechanical damage followed by secondary inflammatory and excitotoxic cascades.
Ischemic Stroke Recovery
Morris et al. showed that TB4 treatment following embolic stroke in rats improved functional neurological outcomes, increased angiogenesis and neurogenesis in the ischemic boundary zone, and promoted white matter remodeling. The intranasal route is especially relevant for stroke recovery because time-to-treatment is critical and intranasal administration is non-invasive, does not require IV access, and can be initiated rapidly in pre-hospital settings. Preclinical studies with intranasal peptide delivery in stroke models have demonstrated rapid brain uptake within 30-60 minutes of administration.
Intranasal Peptide Delivery to the Brain
The nose-to-brain pathway has been extensively characterized for therapeutic peptide delivery. Lochhead and Thorne (2012) reviewed the anatomical and physiological basis of intranasal CNS delivery, establishing that molecules deposited in the upper nasal cavity can reach the brain within minutes via extracellular transport along olfactory and trigeminal nerves. Key factors affecting brain bioavailability include molecular weight (TB4 at ~5 kDa is within the favorable range), formulation characteristics, and deposition pattern in the nasal cavity. Mucoadhesive formulations and absorption enhancers can further improve CNS delivery of intranasally administered peptides.
Neuroinflammation and Microglial Modulation
TB4 modulates neuroinflammatory responses by shifting microglial polarization from the pro-inflammatory M1 phenotype toward the anti-inflammatory M2 phenotype. Sosne et al. (2007) showed that TB4 suppresses NF-kappaB signaling and reduces production of TNF-alpha and IL-1beta. In brain injury models, this translates to reduced secondary damage from neuroinflammation, preservation of the blood-brain barrier integrity around the injury site, and improved functional outcomes.
Anti-Inflammatory Properties
Young et al. (1999) established that TB4-SO inhibits the production of pro-inflammatory cytokines by monocytes stimulated with lipopolysaccharide (LPS). Unlike TB-4, which has modest anti-inflammatory effects mediated through actin regulation and NF-kappaB modulation, TB4-SO acts through a distinct pathway involving glucocorticoid receptor potentiation. This makes TB4-SO more potent as an anti-inflammatory agent despite lacking all cytoskeletal activity.
Sosne et al. (2004) compared the anti-inflammatory effects of TB-4 and TB4-SO in corneal epithelial cell models and found that both reduce inflammatory mediator expression, but through different signaling cascades. TB4-SO's effects were not blocked by actin-disrupting agents, confirming mechanism independence from cytoskeletal regulation.
Innate Immune Response
TB4-SO plays a role in the resolution phase of innate immunity. During acute inflammation, neutrophils and macrophages generate reactive oxygen species that oxidize local TB-4 to TB4-SO. The resulting TB4-SO then acts as an endogenous brake on the inflammatory response. Huff et al. (2001) identified TB4-SO in human wound fluid, establishing its presence at physiologically relevant concentrations in inflammatory environments.
Glucocorticoid Sensitization
Huang et al. (2006) demonstrated that TB4-SO enhances glucocorticoid receptor (GR) nuclear translocation and transcriptional activity in immune cells. This sensitization mechanism means that TB4-SO amplifies the effects of endogenous cortisol without requiring exogenous steroid administration, potentially offering anti-inflammatory benefits without the side effects of glucocorticoid therapy.
Carbon Tetrachloride Injury Models
The carbon tetrachloride (CCl4) model is the most widely used experimental system for studying hepatic fibrosis, and TB-4 has been extensively evaluated in this context. Chronic CCl4 administration to rodents produces progressive pericentral fibrosis that mimics human alcoholic and toxin-induced liver disease.
Sosne et al. (2015) demonstrated that TB-4 treatment in CCl4-injured mice significantly reduced collagen deposition (measured by Sirius Red staining), alpha-SMA expression, and hydroxyproline content compared to vehicle-treated controls. The anti-fibrotic effect was observed both when TB-4 was administered concurrently with CCl4 (prevention) and after fibrosis was already established (reversal), with the reversal finding carrying particular clinical significance.
Kim et al. investigated TB-4's dose-response relationship in the CCl4 model and found that intraperitoneal TB-4 at 5-15 mg/kg produced dose-dependent reductions in hepatic collagen, with histological improvement visible at the lowest dose and near-complete fibrosis reversal at the highest dose after 4 weeks of treatment.
Liver Fibrosis Reversal Mechanisms
The concept of fibrosis reversal — the resolution of established scar tissue — has transformed hepatology over the past two decades. While previously considered irreversible, hepatic fibrosis is now recognized as a dynamic process amenable to therapeutic intervention. TB-4 is among the most effective agents demonstrated to achieve fibrosis reversal in preclinical models.
TB-4-mediated fibrosis reversal involves multiple coordinated processes: deactivation or apoptosis of HSCs (removing the source of excess ECM), upregulation of matrix-degrading enzymes (MMPs), downregulation of MMP inhibitors (TIMPs), and macrophage phenotype switching from pro-fibrotic (M2) to fibrolytic (restorative) phenotypes. This multimodal mechanism distinguishes TB-4 from agents targeting single pathways.
The kinetics of TB-4-mediated fibrosis reversal in rodent models suggest that measurable histological improvement occurs within 2-4 weeks of treatment initiation, with more substantial resolution by 6-8 weeks. Complete architectural restoration (reversal of bridging fibrosis and nodule formation) has been reported in early-stage cirrhosis models but remains incomplete in advanced cirrhosis.
Hepatocyte Proliferation and Liver Regeneration
In addition to its anti-fibrotic properties, TB-4 actively promotes hepatocyte proliferation, supporting the regenerative arm of liver repair. This dual activity is mechanistically important because fibrosis resolution without concurrent hepatocyte regeneration leaves behind necrotic areas and impaired liver function.
TB-4 stimulates hepatocyte proliferation through several pathways, including HGF/c-Met activation, Wnt/beta-catenin signaling, and direct mitogenic effects. In partial hepatectomy models, exogenous TB-4 administration accelerated liver mass restoration and enhanced BrdU incorporation in hepatocytes. In CCl4 models, TB-4-treated animals showed increased hepatocyte proliferation (Ki67-positive cells) alongside decreased fibrosis markers.
The connection between TB-4 and hepatocyte growth factor (HGF) signaling is particularly noteworthy. TB-4 appears to upregulate HGF expression in hepatic mesenchymal cells, creating a paracrine loop that promotes hepatocyte survival and proliferation. This crosstalk between TB-4's direct anti-fibrotic effects and its indirect pro-regenerative effects via HGF represents a therapeutically advantageous mechanism.
Wound Healing and Tissue Repair
The most extensively studied property of TB-500 is its ability to accelerate wound healing. Research in multiple animal models has demonstrated that TB-500 promotes full-thickness wound closure, increases the rate of re-epithelialization, and enhances collagen deposition at wound sites. Malinda et al. (1999) showed that topical or systemic administration of TB4 accelerated dermal wound healing in aged mice, with treated wounds showing significantly increased collagen deposition and angiogenesis compared to controls. The peptide promotes migration of keratinocytes and endothelial cells to the wound bed, increases angiogenesis within the wound, and reduces scar formation through improved collagen organization.
Philp et al. (2004) further demonstrated that TB4 promotes wound healing through multiple mechanisms: increasing cell migration, accelerating collagen deposition, and promoting angiogenesis. In full-thickness excisional wound models in rats, TB4-treated wounds showed 42% faster closure rates and significantly improved cosmetic outcomes with reduced scarring.
Neuroprotection
Emerging research demonstrates that TB-500 has significant neuroprotective properties. Xiong et al. (2012) showed in a rat model of traumatic brain injury (TBI) that intravenous TB4 treatment reduced neuronal cell death, promoted oligodendrocyte differentiation, and enhanced axonal remodeling. The peptide also promoted neuroblast migration from the subventricular zone to areas of cortical injury.
Morris et al. (2010) demonstrated that TB4 treatment following embolic stroke in rats improved functional neurological outcomes, increased angiogenesis and neurogenesis in the ischemic boundary zone, and promoted white matter remodeling. These findings suggest TB4 activates multiple neuroregenerative pathways beyond simple neuroprotection.
Anti-Inflammatory Effects
TB-500 has demonstrated significant anti-inflammatory activity across multiple organ systems. Sosne et al. (2007) showed that in corneal injury models, TB4 reduced inflammatory cell infiltration, decreased levels of pro-inflammatory cytokines (TNF-alpha, IL-1beta, MIP-2), and promoted anti-inflammatory mediator production. These effects appear to be mediated through suppression of NF-kappaB signaling.
Sosne et al. (2010) further demonstrated that TB4 exerts potent anti-inflammatory effects in dry eye disease models, reducing reactive oxygen species, suppressing NF-kappaB activation, and decreasing matrix metalloproteinase levels, establishing TB4 as a multi-modal anti-inflammatory peptide.
Ocular Surface Repair
TB4 has been extensively studied for corneal wound healing and ocular surface repair. Dunn et al. (2010) reviewed the extensive evidence supporting TB4 in corneal repair, including promotion of corneal epithelial cell migration, reduction of inflammation, and prevention of corneal scarring. RegeneRx Biopharmaceuticals developed RGN-259, a topical TB4 formulation that advanced through clinical trials for dry eye disease and neurotrophic keratopathy. Phase 2 trials demonstrated significant improvements in corneal staining scores and symptom relief.
Musculoskeletal Repair
TB-500 has been extensively studied for its effects on tendon, ligament, and muscle repair. In equine veterinary medicine, TB4 has been used to treat tendon and ligament injuries, with studies reporting accelerated healing and improved structural integrity of repaired tissues.
Ehrlich & Bhatt (2021) reviewed the effects of TB4 on tendon healing, reporting that TB4 treatment increased tendon strength and improved collagen fiber alignment in rat Achilles tendon injury models. In skeletal muscle injury models, TB-500 promotes satellite cell activation and myoblast migration, accelerating muscle fiber regeneration.
Kleinman & Sosne (2004) showed that TB4 stimulates the outgrowth and survival of corneal epithelial cells, demonstrating broader regenerative capabilities beyond musculoskeletal tissues.
Comparison with Other TB-4 Applications
TB-4's hepatic effects share mechanistic features with its established roles in other organ systems but also exhibit important differences:
-
Cardiac repair: In cardiac fibrosis following myocardial infarction, TB-4 promotes angiogenesis, cardiomyocyte survival, and anti-fibrotic effects. The hepatic context differs in that the liver's endogenous regenerative capacity is substantially greater than the heart's, and HSCs (rather than cardiac fibroblasts) are the primary fibrogenic cells.
-
Dermal wound healing: TB-4's wound healing properties (the basis for its commercial development as TB-500) involve keratinocyte and endothelial cell migration. In the liver, the migration-promoting effects are redirected toward hepatocyte repopulation and macrophage recruitment for scar resolution.
-
Ocular surface healing: TB-4 (marketed as RGN-259) has advanced furthest in clinical development for corneal wound healing. The parallels with hepatic application include anti-inflammatory and anti-fibrotic mechanisms, but the delivery route and pharmacokinetic requirements differ substantially.
Clinical Translation Challenges
Despite robust preclinical evidence, clinical translation of TB-4 for liver disease faces several challenges:
-
Delivery and pharmacokinetics: TB-4's half-life of approximately 2 hours necessitates frequent dosing or sustained-release formulations for chronic liver disease treatment. Hepatic first-pass metabolism limits oral bioavailability, favoring parenteral administration.
-
Dose optimization: The optimal dose for hepatic anti-fibrotic effects in humans has not been established. Preclinical doses (5-15 mg/kg in rodents) do not translate linearly to human doses, and the therapeutic window for hepatic versus systemic effects requires characterization.
-
Biomarker development: Demonstrating fibrosis reversal in clinical trials requires validated non-invasive biomarkers. Liver biopsy, the gold standard, is invasive and subject to sampling error. Non-invasive markers (FibroScan, ELF score, serum biomarker panels) must be validated specifically for TB-4 response assessment.
-
Patient selection: The heterogeneity of liver fibrosis etiology (viral, alcoholic, metabolic, autoimmune) creates challenges for trial design. TB-4's broad anti-fibrotic mechanism suggests potential efficacy across etiologies, but this must be demonstrated empirically.
-
Regulatory pathway: No anti-fibrotic drug has been approved specifically for liver fibrosis, creating regulatory uncertainty. TB-4's clinical development in ophthalmology (RGN-259) provides safety data that may facilitate hepatic indications.
Sepsis and Systemic Inflammation
TB4-SO has demonstrated significant protective effects in animal models of sepsis. Badamchian et al. (2003) showed that TB4-SO administration improves survival in a cecal ligation and puncture (CLP) model of polymicrobial sepsis in mice. Treated animals showed reduced serum TNF-alpha levels, decreased neutrophil infiltration into organs, and improved hemodynamic stability compared to controls. The peptide was effective when administered both prophylactically and therapeutically after sepsis onset.
Hepatic Stellate Cell Biology
The hepatic stellate cell is the central effector cell of liver fibrosis and the primary cellular target of TB-4 in the hepatic context. In the normal liver, quiescent HSCs reside in the space of Disse and store retinyl esters (vitamin A). Upon liver injury, HSCs undergo activation, a process involving loss of retinoid stores, proliferation, contractility, and massive upregulation of ECM production.
Barnaeva et al. (2007) provided the foundational in vitro evidence that TB-4 directly inhibits HSC activation. Using primary rat HSCs cultured on plastic (which spontaneously activates HSCs, mimicking in vivo activation), TB-4 treatment reduced alpha-SMA expression by 40-60%, decreased collagen type I secretion, and inhibited HSC proliferation in a dose-dependent manner. Critically, TB-4 also promoted apoptosis of activated HSCs while sparing quiescent cells, suggesting selectivity for the pathological phenotype.
Subsequent studies by Shah et al. (2013) demonstrated that TB-4's effects on HSCs extend beyond simple inhibition of activation. TB-4 can induce reversion of fully activated myofibroblastic HSCs back to a quiescent-like state, characterized by re-accumulation of lipid droplets, downregulation of activation markers, and restored vitamin A storage. This reversion phenotype is distinct from HSC apoptosis and represents an alternative mechanism for fibrosis resolution.
TGF-beta/Smad Pathway Interactions
Transforming growth factor-beta 1 (TGF-beta1) is the dominant profibrogenic cytokine in the liver. It drives HSC activation, ECM production, and epithelial-mesenchymal transition through canonical Smad signaling (Smad2/3 phosphorylation) and non-canonical pathways (MAPK, PI3K/Akt).
TB-4 modulates TGF-beta signaling at multiple levels. Direct Smad interference involves reduction of Smad2/3 phosphorylation, decreasing transcriptional activation of profibrotic genes including collagen I/III, fibronectin, and CTGF. Simultaneously, TB-4 upregulates Smad7, the endogenous inhibitory Smad that creates a negative feedback loop on TGF-beta receptor signaling.
Beyond Smad modulation, TB-4 reduces TGF-beta1 production by macrophages and hepatocytes in the injured liver, addressing the upstream signal that initiates fibrogenic cascades. This dual action — reducing both the ligand and its downstream signaling — may explain TB-4's robust anti-fibrotic efficacy relative to agents targeting only one level of the TGF-beta pathway.
Cardiac Protection and Repair
TB-500 has shown remarkable cardioprotective properties in animal models of myocardial infarction. Bock-Marquette et al. (2004) demonstrated in a landmark Nature study that systemic administration of TB4 prior to coronary artery ligation in mice significantly reduced infarct size and improved cardiac function. The peptide activated the survival kinase Akt (protein kinase B), promoting cardiomyocyte survival following ischemic injury.
Smart et al. (2007) showed that TB4 can reactivate the epicardial developmental program in adult mouse hearts, stimulating epicardium-derived progenitor cells to migrate into the myocardium and differentiate into cardiomyocytes. Smart et al. (2011) later demonstrated that TB4 priming of the adult epicardium, followed by myocardial infarction, resulted in de novo cardiomyocyte formation from epicardial progenitors, representing a potential regenerative strategy.
Additional studies have shown that TB4 reduces cardiac fibrosis following myocardial infarction and promotes neovascularization of ischemic myocardium, improving both systolic and diastolic function in treated animals.
Safety Profile
TB-500 has been generally well-tolerated in animal studies and veterinary applications, with no significant adverse effects reported at standard research doses. The peptide does not appear to cause systemic toxicity, organ damage, or immune suppression in preclinical models.
Known safety considerations include:
- Tolerability: No significant adverse effects observed in multiple animal species at therapeutic doses
- Pro-angiogenic concerns: Due to its angiogenic properties, theoretical concerns exist regarding use in the presence of active malignancies, as enhanced blood vessel formation could potentially support tumor growth
- Injection site reactions: Mild, transient injection site discomfort reported in veterinary applications
- Cardiovascular: No adverse cardiovascular effects observed in preclinical cardiac studies
- Long-term data: Long-term safety data in humans remains limited, as most research has been conducted in animal models
Pharmacokinetic Profile
TB-500 — Pharmacokinetic Curve
Subcutaneous injectionOngoing & Future Research
- RegeneRx Biopharmaceuticals has advanced RGN-259 (topical TB4) through Phase 2 trials for dry eye disease and neurotrophic keratopathy.
- Emerging research on TB4 for spinal cord injury repair and neuroinflammation reduction.
- Investigation of TB4-loaded hydrogels and nanoparticle delivery systems for sustained release at wound sites.
- Growing interest in TB4's role in stem cell mobilization and cardiac regenerative medicine.
Quick Start
- Typical Dose
- 2-5 mg
- Frequency
- 2x weekly
- Route
- Subcutaneous injection
- Timing
- Flexible — no fasting or specific timing required
- Cycle Length
- 6-8 weeks (loading), then maintenance as needed
- Break Between Cycles
- 4-6 weeks
- Storage
- Refrigerate 2-8°C; use reconstituted solution within 28 days
Molecular Structure
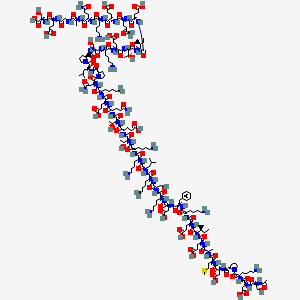
- Formula
- C212H350N56O78S
- Weight
- 4963.44 Da
- Length
- 43 amino acids
- CAS
- 77591-33-4
- PubChem CID
- 16132341
- Exact Mass
- 4962.4930 Da
- LogP
- -43.5
- TPSA
- 2250 Ų
- H-Bond Donors
- 72
- H-Bond Acceptors
- 88
- Rotatable Bonds
- 180
- Complexity
- 12200
Identifiers (SMILES, InChI)
InChI=1S/C212H350N56O78S/c1-16-106(7)166(261-190(323)131(64-75-160(292)293)231-172(305)109(10)228-174(307)134(78-91-347-15)245-196(329)140(98-165(302)303)255-201(334)147-53-39-88-266(147)209(342)135(52-29-38-87-221)250-197(330)139(97-164(300)301)254-198(331)143(100-269)229-113(14)276)204(337)246-129(62-73-158(288)289)187(320)233-118(47-24-33-82-216)179(312)252-137(94-114-42-19-18-20-43-114)194(327)253-138(96-163(298)299)195(328)237-121(50-27-36-85-219)181(314)258-144(101-270)199(332)239-119(48-25-34-83-217)178(311)251-136(92-104(3)4)193(326)236-116(45-22-31-80-214)175(308)235-122(51-28-37-86-220)189(322)263-168(110(11)273)207(340)249-133(66-77-162(296)297)192(325)264-169(111(12)274)206(339)248-126(58-69-152(224)279)184(317)243-128(61-72-157(286)287)186(319)234-120(49-26-35-84-218)180(313)256-142(95-153(225)280)211(344)268-90-40-54-148(268)202(335)257-141(93-105(5)6)210(343)267-89-41-55-149(267)203(336)259-145(102-271)200(333)238-117(46-23-32-81-215)177(310)244-132(65-76-161(294)295)191(324)265-170(112(13)275)208(341)262-167(107(8)17-2)205(338)247-130(63-74-159(290)291)188(321)241-125(57-68-151(223)278)183(316)242-127(60-71-156(284)285)185(318)232-115(44-21-30-79-213)176(309)240-124(56-67-150(222)277)173(306)227-108(9)171(304)226-99-154(281)230-123(59-70-155(282)283)182(315)260-146(103-272)212(345)346/h18-20,42-43,104-112,115-149,166-170,269-275H,16-17,21-41,44-103,213-221H2,1-15H3,(H2,222,277)(H2,223,278)(H2,224,279)(H2,225,280)(H,226,304)(H,227,306)(H,228,307)(H,229,276)(H,230,281)(H,231,305)(H,232,318)(H,233,320)(H,234,319)(H,235,308)(H,236,326)(H,237,328)(H,238,333)(H,239,332)(H,240,309)(H,241,321)(H,242,316)(H,243,317)(H,244,310)(H,245,329)(H,246,337)(H,247,338)(H,248,339)(H,249,340)(H,250,330)(H,251,311)(H,252,312)(H,253,327)(H,254,331)(H,255,334)(H,256,313)(H,257,335)(H,258,314)(H,259,336)(H,260,315)(H,261,323)(H,262,341)(H,263,322)(H,264,325)(H,265,324)(H,282,283)(H,284,285)(H,286,287)(H,288,289)(H,290,291)(H,292,293)(H,294,295)(H,296,297)(H,298,299)(H,300,301)(H,302,303)(H,345,346)/t106-,107-,108-,109-,110+,111+,112+,115-,116-,117-,118-,119-,120-,121-,122-,123-,124-,125-,126-,127-,128-,129-,130-,131-,132-,133-,134-,135-,136-,137-,138-,139-,140-,141-,142-,143-,144-,145-,146-,147-,148-,149-,166-,167-,168-,169-,170-/m0/s1
UGPMCIBIHRSCBV-XNBOLLIBSA-NResearch Indications
Tissue Repair
Accelerated healing of muscle fibers with reduced recovery time via enhanced cell migration and satellite cell activation.
Connective tissue repair with reduced scar formation and improved biomechanical properties. Widely used in equine veterinary medicine.
Enhanced dermal closure, improved angiogenesis, and reduced inflammation. Phase 2 trials showed accelerated healing of chronic pressure and venous stasis ulcers.
Protects bone marrow mesenchymal stem cells from oxidative stress and maintains osteogenic differentiation capacity.
Cardiovascular
Reduces infarct size and improves cardiac function through Akt activation and cardiomyocyte survival. Landmark Nature study demonstrated significant benefits.
Reactivates epicardial developmental program, stimulating de novo cardiomyocyte formation from epicardial progenitors.
Reduces fibrosis following myocardial infarction and promotes neovascularization of ischemic myocardium.
Neurological
Reduces neuronal cell death, promotes oligodendrocyte differentiation, and enhances axonal remodeling following TBI.
Improves functional neurological outcomes, increases angiogenesis and neurogenesis in ischemic boundary zone.
Reduces neuroinflammation, promotes axonal regeneration, and improves locomotor function in preclinical models.
Ocular
Phase 2 clinical trials of RGN-259 (topical TB4) demonstrated significant improvements in corneal staining scores and symptom relief.
Promotes corneal epithelial cell migration, reduces inflammation, and prevents corneal scarring.
Athletic Recovery
Faster recovery from intense training with reduced muscle soreness and improved tissue repair.
Enhanced repair mechanisms may reduce injury risk through tissue remodeling.
Hepatic Fibrosis
Preclinical evidence shows Tβ4 protects hepatocytes by inhibiting GPX4-mediated ferroptosis, reducing liver fibrosis and inflammation in NAFLD models.
Tβ4 prevents oxidative stress, inflammation, and fibrosis in ethanol-induced liver injury by suppressing MeCP2 and reversing PPARγ expression.
Exogenous Tβ4 significantly attenuates pathological changes in CCl4-induced acute liver injury and fibrosis in rodent models via MAPK/NF-κB and Notch signaling suppression.
Hepatic Regeneration
Tβ4 modulates hepatic stellate cell activation via Hedgehog signaling, a key mechanism in liver fibrosis progression and resolution.
Exogenous Tβ4 promotes hepatic tissue regeneration through anti-inflammatory and anti-apoptotic pathways, with TGF-β receptor downregulation reducing fibrogenic gene expression.
Research Protocols
oral
Hepatic first-pass metabolism limits oral bioavailability, favoring parenteral administration.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Rodents | 5-15 mg | Per protocol | — |
intravenous Injection
Administered via intravenous injection.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Rodents | 5-15 mg | Per protocol | — |
intranasal Injection
Intranasal thymosin beta-4 (TB4) represents a specialized delivery approach for the 43-amino acid tissue repair protein, utilizing the nose-to-brain pathway to bypass the blood-brain barrier (BBB) and achieve direct central nervous system distribution. The intranasal route circumvents this limitatio
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Dosing (preclinical) | 1-10 µg, 6 mg | Daily | —(Route: Intranasal) |
intraperitoneal Injection
Comparison with Systemic TB-500 Administration Systemic TB-500 (subcutaneous or intraperitoneal injection) distributes broadly across peripheral tissues and crosses the BBB to a limited extent.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| CCl | 5-15 mg | Per protocol | 4 weeks |
| Neuroprotection used | 6 mg | Per protocol | — |
intramuscular Injection
Alternative injection route, particularly for larger muscle groups. Same dosing as subcutaneous.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Muscle injury recovery | 2-5 mg | 2-3x weekly | 6-8 weeks(Inject into or near affected muscle group) |
topical
Topical formulations used in clinical research for wound healing and ocular conditions. RGN-259 is a topical TB4 formulation that advanced through Phase 2 clinical trials.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Chronic wound healing | 0.02-0.03% concentration | 2x daily | — |
| Acute wound healing | 0.01-0.02% concentration | 2x daily | — |
| Dry eye (RGN-259) | 0.1% concentration eye drops | 1-2x daily | 28-56 days |
subcutaneous Injection
Standard subcutaneous injection protocol with gradual titration. Inject abdominally or near injury site.
| Goal | Dose | Frequency | Duration |
|---|---|---|---|
| Loading phase | 500 mcg | Once daily | Weeks 1-2 |
| Escalation | 600 mcg | Once daily | Weeks 3-4 |
| Standard dose | 750 mcg | Once daily | Weeks 5-8 |
| Full dose | 1,000 mcg | Once daily | Weeks 9-12(Target ~5 mg/week. Titrate up ~100-150 mcg every 2 weeks.) |
Reconstitution Guide (5mg vial + 3mL BAC water)
- Wipe vial tops with alcohol swab
- Draw 3.0 mL bacteriostatic water into syringe
- Inject slowly down the inside wall of the peptide vial
- Gently swirl to dissolve — never shake
- Resulting concentration: 1.67 mg/mL
- For 500 mcg dose: draw 30 units (0.30 mL)
- For 750 mcg dose: draw 45 units (0.45 mL)
- For 1,000 mcg dose: draw 60 units (0.60 mL)
- Store reconstituted vial refrigerated at 2-8°C
Interactions
Peptide Interactions
The most popular peptide healing stack ('Wolverine Stack'). TB-500 enhances cell migration via actin sequestration while BPC-157 drives angiogenesis via VEGFR2. Together they address both vascular supply and cellular repair simultaneously.
TB-500 promotes cell motility and ECM remodeling while GHK-Cu stimulates collagen synthesis and gene expression. Complementary tissue regeneration mechanisms.
Combined tissue repair with GH-releasing effects for comprehensive recovery support.
Enhanced muscle and tissue regeneration through complementary growth pathways.
The most popular research combination. TB-500 enhances cell migration via actin sequestration while BPC-157 drives angiogenesis via VEGFR2. Together they address both vascular supply and cellular repair simultaneously. See BPC-157/TB-500 Blend.
Both peptides suppress NF-kappaB-mediated inflammation through different upstream mechanisms. KPV acts intracellularly via PepT1 uptake; TB4-SO acts via glucocorticoid receptor sensitization. Non-overlapping mechanisms suggest additive potential.
Both thymus-derived peptides. TB-500 handles tissue repair while Thymosin Alpha-1 modulates immune function. Theoretical synergy for post-surgical recovery.
May enhance tissue sensitivity to growth hormone effects. No negative interactions.
No known negative interactions; potential enhanced recovery.
Different receptor families with no pathway overlap.
TB-4's hepatic effects share mechanistic features with its established roles in other organ systems but also exhibit important differences: - Cardiac repair: In cardiac fibrosis following myocardial infarction, TB-4 promotes angiogenesis, cardiomyocyte survival, and anti-fibrotic effects. The hep...
What to Expect
What to Expect
Initial reduction in inflammation; mild injection site reactions may occur
Reduced inflammation and improved tissue sensation; early cell migration and angiogenesis begin
Accelerated healing rate and improved tissue quality; noticeable pain reduction at injury sites
Maximum tissue repair and regenerative benefits; significant improvement in mobility and function
Continued improvement in tissue strength and function; extended protocols for severe or chronic injuries
Safety Profile
Common Side Effects
- Mild injection site redness or irritation (transient)
- Temporary head rush after injection
- Mild lethargy or fatigue during initial healing phase
Contraindications
- Active cancer or history of cancer (angiogenic properties may theoretically promote tumor vascularization)
- Pregnancy or breastfeeding
- Active treatment with immunosuppressive medications (consult provider)
- Children under 18
- WADA prohibited substance — banned in competitive athletics
Discontinue If
- Persistent or worsening injection site infections
- Severe allergic reaction (rash, swelling, difficulty breathing)
- Unusual prolonged swelling or redness
- Unexpected systemic symptoms
- Any concerning health changes
Drug Interactions
- May interact with anticoagulants due to angiogenic effects
- Potential interaction with immunosuppressants
- Theoretical concern with active cancer treatments due to pro-angiogenic properties
Uncommon
- Headache
- Temporary dizziness
- Mild nausea
Quality Indicators
What to look for
- White, fine lyophilized powder with light fluffy texture
- Clear, colorless solution after reconstitution with no particles
- Proper vacuum seal intact with secure rubber stopper
- Comes with third-party purity certificate (HPLC >=98%)
- Sealed under vacuum or nitrogen atmosphere
Caution
- Slight powder clumping (acceptable if completely dissolves with gentle swirling)
- Powder appears loose rather than solid puck
Red flags
- Yellow or brown discoloration (indicates degradation)
- Crystalline or gel-like consistency
- Persistent cloudiness, floating particles, or precipitation after reconstitution
- Damaged vial seal or contamination signs
- No COA or purity testing available
- Stored/shipped without cold chain
Frequently Asked Questions
References (18)
- [14]Ehrlich HP, Bhatt A. Thymosin beta-4 effects on tendon healing Annals of Anatomy (2021)
→ TB4 increased tendon strength and improved collagen fiber alignment in Achilles tendon injury models
- [16]Wei X, et al. Thymosin beta 4 protects against oxidative stress-induced impairment of osteogenic differentiation of bone marrow-derived mesenchymal stem cells Stem Cell Research & Therapy (2023)
→ TB4 protects bone marrow MSCs from oxidative stress and maintains osteogenic differentiation capacity
- [17]Xing Y, et al. Thymosin beta-4 promotes angiogenesis and wound healing through the PI3K/AKT/eNOS signaling pathway Journal of Cytotherapy (2023)
→ Identified PI3K/AKT/eNOS as key mediator of TB4-induced angiogenesis
- [18]Li Y, et al. Thymosin beta 4 attenuates neuroinflammation and promotes functional recovery after traumatic spinal cord injury International Immunopharmacology (2023)
→ TB4 reduced neuroinflammation, promoted axonal regeneration, and improved locomotor function after SCI
- [2]Grant DS, Rose W, Yaen C, et al. Thymosin beta4 enhances endothelial cell differentiation and angiogenesis Angiogenesis (1999)
→ TB4 promotes angiogenesis both in vitro and in the chick chorioallantoic membrane assay
- [3]Malinda KM, Sidhu GS, Mani H, et al. Thymosin beta4 accelerates wound healing Journal of Investigative Dermatology (1999)
→ Topical or systemic TB4 accelerated dermal wound healing in aged mice with increased collagen and angiogenesis
- [1]Safer D, Elzinga M, Nachmias VT. Thymosin beta 4 and Fx, an actin-sequestering peptide, are indistinguishable Journal of Biological Chemistry (1997)
→ Determined crystal structure of TB4-actin complex, revealing peptide wraps around actin monomer to prevent polymerization
- [4]Philp D, Huff T, Gho YS, et al. The actin binding site on thymosin beta4 promotes angiogenesis FASEB Journal (2004)
→ TB4 promotes wound healing through multiple mechanisms with 42% faster closure rates
- [15]Maar K, et al. Evaluation of Thymosin Beta 4 for the treatment of dry eye disease in a controlled adverse environment Clinical Ophthalmology (2022)
→ RGN-259 significantly improved corneal fluorescein staining and ocular discomfort vs placebo
- [5]Bock-Marquette I, Saxena A, White MD, et al. Thymosin beta4 activates integrin-linked kinase and promotes cardiac cell migration, survival and cardiac repair Nature (2004)
→ Landmark study: systemic TB4 reduced infarct size and improved cardiac function via Akt activation
- [6]Kleinman HK, Sosne G. Thymosin beta 4 promotes dermal healing Vitamins and Hormones (2004)
→ TB4 stimulates outgrowth and survival of corneal epithelial cells
- [7]Smart N, Risebro CA, Melville AA, et al. Thymosin beta4 induces adult epicardial progenitor mobilization and neovascularization Nature (2007)
→ TB4 reactivates the embryonic epicardial developmental program in adult hearts
- [8]Sosne G, Qiu P, Goldstein AL, Wheater M. Biological activities of thymosin beta4 defined by active sites in short peptide sequences FASEB Journal (2007)
→ TB4 modulates inflammatory mediators in corneal epithelial cells via NF-kappaB suppression
- [9]Sosne G, et al. Thymosin beta 4 promotes corneal wound healing and decreases inflammation in vivo following alkali injury Experimental Eye Research (2010)
→ TB4 reduces ROS, suppresses NF-kappaB, and decreases MMP levels in dry eye models
- [10]Morris DC, Cui Y, Cheung WL, et al. A dose-response study of thymosin beta4 for the treatment of acute stroke Journal of Neurological Sciences (2010)
→ TB4 improved functional outcomes and increased angiogenesis/neurogenesis after embolic stroke in rats
- [11]Dunn SP, et al. Thymosin beta 4 in corneal wound healing Annals of the New York Academy of Sciences (2010)
→ Comprehensive review supporting TB4 in corneal repair including cell migration and scar prevention
- [12]Smart N, Bollini S, Dube KN, et al. De novo cardiomyocytes from within the activated adult heart after injury Nature (2011)
→ TB4 priming results in de novo cardiomyocyte formation from epicardial progenitors
- [13]Xiong Y, Mahmood A, Chopp M. Thymosin beta4 treatment for traumatic brain injury Neural Regeneration Research (2012)
→ TB4 reduced neuronal cell death and promoted oligodendrocyte differentiation after TBI
TB-4 Fragment (Ac-SDKP)
Ac-SDKP (N-acetyl-seryl-aspartyl-lysyl-proline) is a naturally occurring tetrapeptide released from thymosin beta-4 by prolyl oligopeptidase. It is a potent anti-fibrotic agent with significant research in cardiac and renal fibrosis, hematopoietic stem cell regulation, and inflammation.
Tesamorelin
Tesamorelin is a synthetic growth hormone-releasing hormone (GHRH) analogue approved under the brand name Egrifta for the reduction of excess abdominal fat in HIV-infected patients with lipodystrophy.